

Risk Factors Associated With Transition From
Acute to Chronic Low Back Pain in US
Patients Seeking Primary CareThis section is compiled by Frank M. Painter, D.C.
Send all comments or additions to: Frankp@chiro.org




FROM: JAMA Network Open 2021 (Feb 1); 4 (2): e2037371 ~ FULL TEXT
OPEN ACCESS Joel M. Stevans, DC, PhD; Anthony Delitto, PT, PhD; Samannaaz S. Khoja, PT, PhD; Charity G. Patterson, PhD et al.
School of Health and Rehabilitation Sciences,
University of Pittsburgh,
Pittsburgh, Pennsylvania
Importance: Acute low back pain (LBP) is highly prevalent, with a presumed favorable prognosis; however, once chronic, LBP becomes a disabling and expensive condition. Acute to chronic LBP transition rates vary widely owing to absence of standardized operational definitions, and it is unknown whether a standardized prognostic tool (ie, Subgroups for Targeted Treatment Back tool [SBT]) can estimate this transition or whether early non-guideline concordant treatment is associated with the transition to chronic LBP.
Objective: To assess the associations between the transition from acute to chronic LBP with SBT risk strata; demographic, clinical, and practice characteristics; and guideline nonconcordant processes of care.
Design, setting, and participants: This inception cohort study was conducted alongside a multisite, pragmatic cluster randomized trial. Adult patients with acute LBP stratified by SBT risk were enrolled in 77 primary care practices in 4 regions across the United States between May 2016 and June 2018 and followed up for 6 months, with final follow-up completed by March 2019. Data analysis was conducted from January to March 2020.
Exposures: SBT risk strata and early LBP guideline nonconcordant processes of care (eg, receipt of opioids, imaging, and subspecialty referral).
Main outcomes and measures: Transition from acute to chronic LBP at 6 months using the National Institutes of Health Task Force on Research Standards consensus definition of chronic LBP. Patient demographic characteristics, clinical factors, and LBP process of care were obtained via electronic medical records.
Results: Overall, 5233 patients with acute LBP (3029 [58%] women; 4353 [83%] White individuals; mean [SD] age 50.6 [16.9] years; 1788 [34%] low risk; 2152 [41%] medium risk; and 1293 [25%] high risk) were included. Overall transition rate to chronic LBP at six months was 32% (1666 patients). In a multivariable model, SBT risk stratum was positively associated with transition to chronic LBP (eg, high-risk vs low-risk groups: adjusted odds ratio [aOR], 2.45; 95% CI, 2.00–2.98; P < .001). Patient and clinical characteristics associated with transition to chronic LBP included obesity (aOR, 1.52; 95% CI, 1.28–1.80; P < .001); smoking (aOR, 1.56; 95% CI, 1.29–1.89; P < .001); severe and very severe baseline disability (aOR, 1.82; 95% CI, 1.48–2.24; P < .001 and aOR, 2.08; 95% CI, 1.60–2.68; P < .001, respectively) and diagnosed depression/anxiety (aOR, 1.66; 95% CI, 1.28–2.15; P < .001). After controlling for all other variables, patients exposed to 1, 2, or 3 nonconcordant processes of care within the first 21 days were 1.39 (95% CI, 1.21–2.32), 1.88 (95% CI, 1.53–2.32), and 2.16 (95% CI, 1.10–4.25) times more likely to develop chronic LBP compared with those with no exposure (P < .001).
Conclusions and relevance: In this cohort study, the transition rate to chronic LBP was substantial and increased correspondingly with SBT stratum and early exposure to guideline nonconcordant care.
From the FULL TEXT Article:
Introduction
Low back pain (LBP) is the leading cause of disability in the United States, annually accounting for 4.3 million years lived with disability, nearly twice the burden of any other health condition. [1] Overall, 13% of adults have chronic LBP, with one-third experiencing moderate- to high-impact chronic pain. [2, 3] In the United States, treatment for LBP and related spine disorders now represents the most expensive medical problem, with most costs accrued in ambulatory care settings, including primary care. [4, 5] Chronic LBP contributes most to long-term disability, morbidity, health care, and societal costs, while, acute LBP is given less attention because patients are generally considered to have a favorable prognosis. [6, 7]
Recent evidence has questioned the prevailing belief that acute LBP resolves within 3 months. [8, 9] A systematic review indicated that 2% to 48% (median, 26%) of patients with acute LBP in primary care settings transition to chronic LBP. [10] Wide variability can be attributed to heterogeneous populations and varying operational definitions of acute and chronic LBP. Among a cohort of 605 primary care patients with acute LBP, 9% to 35% were found to have chronic LBP at 6 months depending on chronic LBP operational definitions. [8] Lacking a standardized definition, research investigating determinants and interventions associated with the transition from acute to chronic LBP is hampered, a main factor that led to convening a National Institute of Health (NIH) Task Force Pain Consortium to develop a standardized definition and research standards for chronic LBP. [11]
The NIH Task Force also recommended further study of prognostic instruments, such as the Subgroups for Targeted Treatment (STarT) Back tool (SBT). [11] The SBT is a 9–item instrument designed to identify patients with LBP at risk of persistent functional limitations but has not been investigated to assess the transition to chronic LBP. It is also used to guide treatment decisions, whereby minimal care is provided for low-risk patients and more intensive treatment is recommended as risk increases. [12, 13] The SBT is reliable and valid for predicting poor functional outcomes; however, the SBT has not been investigated as a prognostic tool for the acute to chronic LBP transition. [14]
Clinical guidelines consistently recommend reassurance (eg, most episodes of acute LBP resolve quickly and have a very low likelihood of serious underlying pathology) and advice to maintain activity as tolerated. [15–17] Recently, nonpharmacologic interventions, such as heat, massage, acupuncture, or spinal manipulation, are recommended as first-line treatment options, while initial use of diagnostic imaging, specialty consultation, and prescription of opioid medications in the absence of red flags (eg, fever, fracture, malignant neoplasms) are not recommended. [17] Nonconcordant care can lead to direct and indirect harm, given that it has been linked with medicalization and unnecessary health care utilization.18,19,20 Accumulating evidence indicates that guideline-concordant care has not been successfully implemented in primary care; however, the association of nonconcordant care with the transition to chronic LBP remains unclear. [21, 22]
To obtain estimates of the transition from acute to chronic LBP, assess the SBT prognostic capabilities, and identify pragmatic factors associated with poor outcomes, we conducted a large multisite inception cohort study. We prospectively enrolled patients with acute LBP who were seen in primary care, administered the SBT at baseline, and assessed for chronic LBP at 6 months using the NIH Task Force definition.
The objectives for this study were to assess the association between risk of acute to chronic LBP transition with(1) baseline SBT risk strata;
(2) patient demographic and clinical characteristics and practice characteristics; and
(3) guideline nonconcordant processes of care.
Methods
Study Design and Setting

Figure 1 A detailed study protocol has been published, [23] and reporting of this manuscript follows the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) reporting guideline for cohort studies. Briefly, we conducted an inception cohort study alongside the multisite, pragmatic (ie, tested in real-world settings), cluster randomized Targeted Interventions to Prevent Chronic Low Back Pain in High-Risk Patients (TARGET) clinical trial (NCT02647658). Patients presenting to primary care clinics with acute LBP were stratified by risk (ie, low, medium, and high) for developing chronic LBP using the SBT. [24] High-risk patients were enrolled in the RCT and cohort studies. Medium-risk and low-risk patients were only enrolled in the cohort study. All patients were assessed for the presence or absence of chronic LBP at baseline and 6 months (Figure 1). Patients were enrolled between May 2016 and June 2018 in 77 primary care practices in 4 US health systems (Pittsburgh, Pennsylvania; Boston, Massachusetts; Salt Lake City, Utah; and Baltimore, Maryland), and follow-up was completed by March 2019.
The study was overseen by 4 institutional review boards. [23] Processes conducted within the primary care clinics were viewed as quality improvement and the 6–month survey as research requiring verbal or written informed consent. Sites had varying approaches to obtaining consent for the 6–month survey. In Boston, Salt Lake City, and Pittsburgh, consent was obtained in conjunction with the 6–month survey, while in Baltimore consent was obtained at baseline.
Data Collection
All study data except the 6–month surveys were sourced from existing data fields in the electronic medical records (EMRs). EMR data were collected using standardized data extraction and secure file transfer protocols facilitated via honest brokers.
Cohort Identification
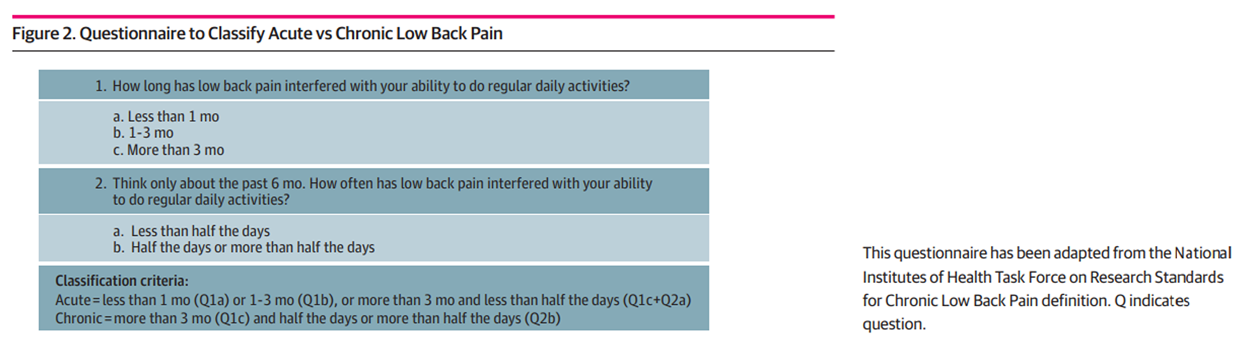
Figure 2 Patients were eligible if they were adults (aged ≥18 years) and presented with a primary concern of acute, bothersome axial LBP or LBP with associated leg pain. To determine the nature of the concern, a 2–item acute/chronic LBP screening questionnaire was created by adapting the NIH Task Force on Research Standards chronic LBP definition.
Patients were considered to have chronic LBP if they reported(1) the presence of pain for more than 3 months and
(2) experienced pain at least half the days in the past 6 months.Those not meeting this definition were classified as having acute LBP (Figure 2). The date the questionnaire was administered was considered the index (ie, baseline) visit. Data were collected via study tablets or verbally by clinical or administrative personnel and uploaded into the EMR. The LBP concern was verified retrospectively using the International Classification of Diseases, Ninth Revision (ICD-9) or ICD-10 Clinical Modification (ICD-10-CM) diagnosis codes from the EMR, and patients were excluded if any diagnoses on the index visit indicated a potential red flag for a serious underlying reason for LBP (eg, fracture, cancer).
Risk Stratification
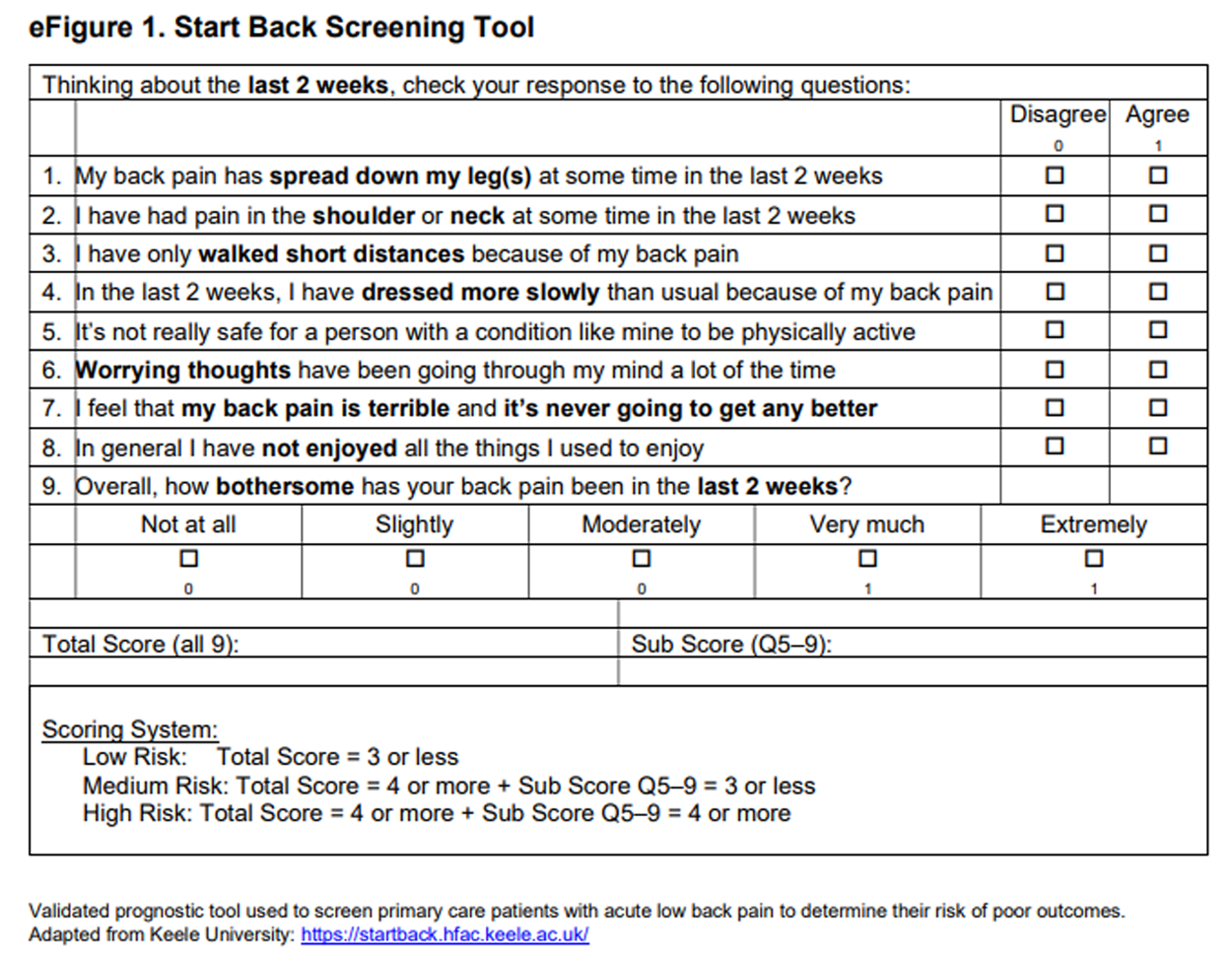
Risk for developing chronic LBP was determined at the index visit using the 9–item version of the SBT. [24] The total score ranges from 0 to 9 and includes a psychological subscale score ranging from 0 to 5. Patients were stratified as low-risk (total score ≤3), medium-risk (total score ≥4 and subscale score ≤3), or high risk (total score ≥4 and subscale score ≥4).24 The SBT was administered in primary care clinics using the same process as the acute/chronic LBP questionnaire (eFigure 1 in the Supplement).
Outcome
Chronic LBP status at 6 months was ascertained by the acute/chronic LBP questionnaire. The survey was collected electronically, by mail, or by telephone by research personnel.
Demographic, Clinical, and Practice Covariates
Patient characteristics included demographic factors (ie, age, sex, race, ethnicity, and health insurance) and clinical characteristics (ie, body mass index, smoking status, LBP diagnosis, psychological comorbidities, self-reported LBP disability). Insurance was collapsed to 4 categories (commercial, Medicare, Medicaid, and other [workers’ compensation, self-pay, missing]). LBP diagnoses and psychological comorbidities were identified via ICD-9 or ICD-10-CM diagnostic codes present at the index visit. LBP disability was assessed via the Oswestry Disability Index (ODI), which was administered at the index visit with the SBT and acute/chronic LBP questionnaire. [25] The ODI scores range from 0 to 100 and were categorized using the following disability definitions: minimal (0–20), moderate (21–40), severe (41–60), very severe (≥61). [25] Practice characteristics included geographic location and the national area deprivation index (ADI). The ADI ranks the Census block or neighborhood in terms of socioeconomic disadvantage. We used the validated Neighborhood Atlas Tool to estimate national level ADI scores for each clinic location. [26]
Nonconcordant Processes of Care
LBP-related processes of care provided by primary care clinicians within 21 days of a patient’s index visit were extracted from the EMR. We used international LBP guidelines and codified these processes of care into 3 categories: pharmacologic therapies, diagnostic imaging, and medical subspecialty referral. [17] We further categorized each process of care as concordant or nonconcordant with these guidelines. Nonconcordant pharmacotherapy was determined using the algorithm provided in eFigure 2 in the Supplement. [17] Briefly, any prescriptions that included opioids were considered nonconcordant. Additionally, prescriptions that included benzodiazepines and/or systemic corticosteroids alone without the presence of nonsteroidal anti-inflammatory drugs or short-term skeletal muscle relaxants were considered nonconcordant. Nonconcordant diagnostic imaging consisted of an order for lumbar radiograph or computed tomography/magnetic resonance imaging (CT/MRI) scan. Nonconcordant medical subspecialty referral included referrals to nonsurgical or surgical specialties (eg, physiatrists, orthopedists, neurologists, neurosurgeons, or pain specialists). To improve interpretation and analysis, we created a composite variable that represented the total count of categories with nonconcordant processes of care. The minimum composite score was 0 (ie, patient received no nonconcordant processes of care), while the maximum composite score was 3 (ie, patients received nonconcordant processes of care in all 3 categories).
Sample Size
Sample size estimates for the high-risk cohort are reported elsewhere. [20] Estimates for low-risk and medium-risk patients were derived from the proportion of patients expected to screen into these 2 strata in the parent trial’s primary care clinics. [23] We assumed a mean of 115 patients would screen as low to medium risk per clinic during enrollment of the trial, with 20% transitioning to chronic LBP, an intracluster correlation of 0.01, and a 60% response rate at 6 months. This provided us at minimum a 79% power to detect 30% relative difference in transition to chronic LBP between patient subgroups (eg, SBT stratum) that could be as small as a 1:4 ratio (ie, the subgroup makes up 20% of the sample compared with the remaining 2 subgroups, which make up 80% of the sample).
Statistical Analysis
We compared baseline demographic, clinical, practice, and process of care characteristics between patients with and without 6–month follow-up surveys using χ2 tests. In these comparisons, we adjusted for clustering at the clinic level using Taylor series linearization for variance estimation. We described the rate of transition to chronic LBP at 6 months for each independent variable across the SBT risk stratum and for all strata combined. Univariate and multivariable associations between the independent variables and transition to chronic LBP were assessed using a generalized linear mixed model with a logit link controlling for the cluster design with a random clinic effect. All variables entered into the multivariable models were categorical and treated as fixed effects with significance of P < .15 for further consideration. All multivariable models controlled for site as a fixed effect. SBT risk strata and the composite nonconcordant process of care score remained in the model, and a backward stepwise elimination with significance set at P < .15 was used to arrive at a final multivariable model. Potential selection bias due to the large proportion of patients with missing 6–month data was addressed by applying stabilized inverse probability weights to the multivariable model. [27] Data analysis was conducted with SAS version 9.4 (SAS Institute). Statistical significance was set at P < .05, and all tests were 2–tailed.
Results

Table 1

Table 2
page 9The final cohort of 5,233 patients with acute LBP and 6–month surveys (Figure 1) were mostly women (3,029 [58%]) and White individuals (4,353 [83%]) with overweight (1,599 [31%]) or obesity (2,308 [44%]). Most patients were diagnosed with axial LBP (3,883 [74%]), and 292 (6%) had an anxiety or depression diagnosis. Risk stratification in the final cohort showed 1,788 (34%) were low risk; 2,152 (41%), medium risk; and 1,293 (25%), high risk for developing chronic LBP. Across risk levels, 1,544 patients (30%) received prescriptions for nonrecommended medications (999 [65%] received opioids); 1,245 (24%) received an radiography or CT/MRI order; and 333 (6%) were referred to a medical specialist (207 [62%] surgeons) within 21 days of the index visit (Table 1). The survey nonresponse rate was 45% (4,314 of 9,547). Nonresponders were less likely to be White individuals, to have overweight or obesity, to not currently smoke, to be prescribed nonconcordant pharmacologic therapies, or to receive care from the Intermountain Healthcare or University of Pittsburgh Medical Center systems (Table 1).
The overall unadjusted acute to chronic LBP transition rate at 6 months was 32% (1,666 of 5,233). The unadjusted rates by low-risk, medium-risk, and high-risk stratum were 19% (333), 33% (703), and 49% (630), respectively. Positive univariate associations were found between chronic LBP at 6 months and exposure to nonconcordant pharmacotherapies (606 of 1,544 [39%] vs 1,060 of 3,689 [29%]; P < .001), diagnostic imaging (447 of 1,245 [36%] vs 1,219 of 3,988 [31%]; P < .001), and referral to medical subspecialists (176 of 333 [53%] vs 1,490 of 4,900 [30%]; P < .001). As the composite nonconcordant processes of care score increased from 0 to 3 , the rates of transition increased in a stepwise fashion from 27% (724 of 2,722) to 53% (20 of 38) (P < .001) (Table 2).
Factors Associated With Transition to Chronic LBP From the Multivariable Model

Table 3
page 10In the multivariable model, SBT risk strata were positively associated with the development of chronic LBP when controlling for all other variables. Compared with patients in the low-risk category, the adjusted odds ratio (aOR) of developing chronic LBP was 2.45 (95% CI, 2.00–2.98) times higher for those in the high-risk category and 1.59 (95% CI, 1.33–1.89) times higher for those in the medium-risk category (P < .001) (Table 3). Furthermore, there was a stepwise linear relationship across each SBT stratum (P for trend < .001).
Baseline disability was positively associated with transition to chronic LBP. The aOR for developing chronic LBP was 1.16 (95% CI, 0.97–1.39) times higher for moderate disability, 1.82 (95% CI, 1.48–2.24) times higher for severe disability, and 2.08 (95% CI, 1.60–2.68) times higher for very severe disability compared with minimal disability (P < .001) (Table 3). Other significant independent factors included health insurance (eg, Medicaid: aOR, 1.91; 95% CI, 1.53–2.38; P < .001), body mass index (eg, obesity: aOR, 1.52; 95% CI, 1.28–1.80; P < .001), smoking status (aOR, 1.56; 95% CI, 1.29–1.89; P < .001), diagnosis at the index visit (back and leg pain: aOR, 1.16; 95% CI, 1.00–1.35; P = .04), and psychological comorbidities (aOR, 1.66; 95% CI, 1.28–2.15; P < .001).
Exposure to nonconcordant care was associated with increased odds of developing chronic LBP (P < .001). The aORs for developing chronic LBP were 1.39 (95% CI, 1.21–2.32), 1.88 (95% CI, 1.53–2.32), and 2.16 (95% CI, 1.10–4.25) times higher for exposure to 1, 2, or 3 nonconcordant processes of care, respectively, compared with 0 nonconcordant processes of care (P < .001) (Table 3). Additionally, there was a positive linear association between the number of nonconcordant processes of care and risk of developing chronic LBP (P for trend = .04).
Inverse Probability Weighted Multivariable Model
The multivariable model was reexamined using stabilized inverse probability weighting, and the results closely matched the direction and magnitude of the aORs in the original model, except for minor differences in the width of the confidence intervals (Table 3). The linear associations between the transition to chronic LBP and SBT strata (P for trend < .001) and the number of nonconcordant processes of care (P for trend = .02) remained significant.
Discussion
We present the results of a large prospective, multicenter study conducted to determine the proportion of patients who transitioned from acute to chronic LBP in primary care settings across 4 geographically dispersed health systems. Based on the NIH Task Force definition, the overall transition to chronic LBP was 32%. The risk of transition was linearly associated with baseline SBT category and whether early care was nonconcordant with current practice guidelines. Collectively, these results indicate that the transition from acute to chronic LBP is much greater than historically appreciated, the SBT can estimate risk of transition, and lack of guideline adherence may increase transition rates. These results expand SBT’s capabilities to include the transition to chronic LBP using the NIH operational definition and reinforce the importance of LBP guidelines.
Practice guidelines do not consistently recommend the use of risk stratification tools, such as the SBT, for acute LBP, likely due to the prevailing perception that acute LBP has a favorable prognosis. [17] The SBT was designed to tailor treatments based on risk of persistent functional limitations. In clinical practice, it may be tempting to focus on high-risk groups given the high transition rate to chronic LBP. However, it should be noted that more than 60% of the 1,666 patients who developed chronic LBP at 6 months were in the low-risk (333 patients) and medium-risk (703) groups. Even though the rate of transition to chronic LBP is lower in these groups, most patients with acute LBP (>75%) fall into these strata. As a result, uniformly applying a minimalist approach (eg, advice, reassurance) to all patients with acute LBP without considering SBT risk status is unwarranted and may lead to suboptimal care. [17] Conversely, uniformly administering resource intensive, multimodal interventions across the entire acute LBP population is unwarranted and may result in low-value care. [5, 28] To ensure appropriate treatment intensity and cost-effectiveness, future research should consider both patient phenotype and the prevalence within each SBT stratum to identify effective and scalable interventions. [29, 30]
In this cohort, patient demographic and clinical factors associated with the chronic LBP transition included obesity, smoking, insurance coverage, LBP with leg pain, baseline disability, and diagnosed depression/anxiety. The role that these factors play in the transition to chronic LBP cannot be ignored; however, many of these factors are difficult to change or nonmodifiable altogether. Importantly, our findings demonstrate that independent of these factors, exposure to nonconcordant processes of care during the early phase of treatment was associated with developing chronic LBP. Nearly half of patients (48%) received at least 1 discordant process of care within 3 weeks of the index visit. Even after controlling for patient characteristics (eg, obesity) and clinical characteristics (eg, baseline disability), increasing numbers of nonconcordant management approaches increased the likelihood of having chronic LBP at 6 months. These rates of nonconcordant processes of care are similar to those reported in a claims analysis from 2.5 million individuals newly diagnosed with LBP. [5] The independent association between nonconcordant care and risk of developing chronic LBP highlights the need to identify strategies that facilitate LBP guideline implementation.
Successful management of LBP is a vexing problem, and health systems have been challenged to develop innovative solutions. [29, 31] Once chronic, LBP is particularly problematic to manage; thus, preventing the transition from acute to chronic LBP is important. Primary care physicians worldwide are under enormous pressure to do more with less, which is the basis for the SBT risk stratification approach. [32] However, LBP guidelines have yet to consistently recommend the use of risk stratification, and implementing this approach in primary care is proving to be difficult. [33, 34] One reason for poor implementation may trace back to physicians’ musculoskeletal training. [35] Medical educators have recognized for years that training in musculoskeletal medicine is suboptimal for medical students, residents, and general practitioners. [35–37] Placing a greater emphasis on a highly prevalent condition, such as LBP, during training may improve implementation at the individual level.
Other possible reasons for poor implementation include high caseloads and the overwhelming volume of guidelines directed at primary care; typical physicians would need an estimated 18 hours per day to address all guideline recommendations. [38] It is time to test supportive models of care to assist primary care practitioners in addressing this substantial public health problem. Evidence from other conditions suggests that organizational strategies that incorporate nonphysician health professionals (eg, nurse practitioners or physician assistants) to comanage cases can improve guideline adherence in primary care. [39] The Primary Spine Practitioner is another model proposed in the United States in which chiropractors and physical therapists serve as the initial or early point of contact for patients with LBP. [40] Another potentially beneficial organizational strategy is the use of multidisciplinary teams comprised of medical specialists and other health professionals (eg, integrated practice units). [41–43] Future studies need to evaluate whether different models of care in conjunction with risk stratification can improve guideline concordance, patient outcomes, and decrease the total cost of care.
Limitations
This study has limitations. We used survey methods to collect 6–month outcomes due to the large sample size and pragmatic nature of the study. Our response rate was 55%, which would be considered low by standards of a clinical efficacy trial. However, response rates in the range of 50% to 60% are considered to pose minimal risk of nonresponse bias when using survey methods. In addition, our inverse probability weighted analysis accounted for selection bias due to nonresponse and confirmed our original conclusions. [27, 44, 45]
In this study, we relied on EMR data to develop the clinical profile of patients with acute LBP. Although widely generalizable, EMR data may not reflect all pertinent clinical findings, conditions, or comorbidities considered or addressed by the physician, nor do they include all potential confounders that may affect the transition to chronic LBP or necessitate deviation from guidelines. For example, nonconcordant care may be provided to patients with more complex acute LBP. We cannot completely rule out residual confounding; however, we controlled for a broad set of factors associated with higher clinical severity (baseline disability and LBP with leg pain) and factors that complicate management (body mass index, smoking, psychological comorbidities, and SBT risk status). [46]
Conclusions
This large inception cohort study found that the transition from acute to chronic LBP was substantial and the SBT was a robust prognostic tool. Early exposure to guideline nonconcordant care was significantly and independently associated with the transition to chronic LBP after accounting for patient demographic and clinical characteristics, such as obesity, smoking, baseline disability, and psychological comorbidities. These findings suggest that an emphasis should be placed on discovering strategies to successfully implement guideline concordant care in the primary care setting to reduce the development of chronic LBP.
Supplementary Material
eFigure 1. Start Back Screening Tool
eFigure 2. Algorithm to Classify Guideline Nonconcordant Care for Pharmacologic Therapies
Funding/Support:
This research work was funded by award PCS-1402-10867 from the Patient-Centered Outcomes Research Institute
Conflict of Interest Disclosures:
Dr Delitto reported receiving grants from the National Institutes of Health outside the submitted work.
Dr Sowa reported receiving grants from the National Institutes of Health outside the submitted work.
Dr George reported receiving grants from the National Institutes of Health and receiving personal fees from Rehab Essentials and MedRisk outside the submitted work.
No other disclosures were reported
Author Contributions:
Dr Stevans had full access to all of the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Concept and design: Delitto, Schneider, Greco, Freel, Sowa, Wasan, Brennan, Hunter, Wegener, Ephraim, Friedman, George, Saper.
Acquisition, analysis, or interpretation of data: Stevans, Delitto, Khoja, Patterson, Smith, Freburger, Greco, Sowa, Wasan, Brennan, Minick, Wegener, Ephraim, Beneciuk, George, Saper.
Drafting of the manuscript: Stevans, Delitto, Khoja, Schneider, Freburger, Freel, Brennan, Beneciuk, George, Saper.
Critical revision of the manuscript for important intellectual content: Stevans, Delitto, Khoja, Patterson, Smith, Schneider, Freburger, Greco, Sowa, Wasan, Brennan, Hunter, Minick, Wegener, Ephraim, Friedman, Beneciuk, George, Saper.
Statistical analysis: Stevans, Patterson, Smith, Schneider, Wasan.
Obtained funding: Delitto, Wasan, Brennan, Ephraim, Saper.
Administrative, technical, or material support: Stevans, Delitto, Khoja, Schneider, Freburger, Greco, Freel, Sowa, Wasan, Brennan, Hunter, Minick, Wegener, Ephraim, Friedman, George.
Supervision: Stevans, Delitto, Sowa, Wasan, Brennan, Ephraim, George, Saper.
References:
Global Burden of Disease Collaborative Network
Global Burden of Disease Study 2017 (GBD 2017) results.
Accessed January 11, 2021.
http://ghdx.healthdata.org/gbd-results-toolVon Korff M, Scher AI, Helmick C, et –al.
United States National Pain Strategy for population research:
concepts, definitions, and pilot data
J Pain. 2016;17(10):1068-1080Dahlhamer J, Lucas J, Zelaya C, et –al.
Prevalence of Chronic Pain and High-Impact Chronic Pain
Among Adults - United States, 2016
MMWR Morb Mortal Wkly Rep. 2018 (Sep 14); 67 (36): 1001-1006Dieleman JL, Cao J, Chapin A, et al.
US Health Care Spending by Payer and Health Condition, 1996-2016
JAMA 2020 (Mar 3); 323 (9): 863–884Kim LH, Vail D, Azad TD, et al.
Expenditures and Health Care Utilization Among Adults
with Newly Diagnosed Low Back and Lower Extremity Pain
JAMA Network Open 2019 (May 3); 2 (5): e193676Pengel LH, Herbert RD, Maher CG, Refshauge KM.
Acute low back pain: systematic review of its prognosis.
BMJ. 2003;327(7410):323.
doi: 10.1136/bmj.327.7410.323Koes BW, van Tulder MW, Thomas S.
Diagnosis and Treatment of Low Back Pain
British Medical Journal 2006 (Jun 17); 332 (7555): 1430–1434Mehling WE, Gopisetty V, Bartmess E, et al..
The prognosis of acute low back pain in primary care in
the United States: a 2-year prospective cohort study.
Spine (Phila Pa 1976). 2012;37(8):678-684.
doi: 10.1097/BRS.0b013e318230ab20Itz CJ, Geurts JW, van Kleef M, Nelemans P.
Clinical Course of Non-specific Low Back Pain: A Systematic
Review of Prospective Cohort Studies Set in Primary Care
European Journal of Pain 2013 (Jan); 17 (1): 5–15Chou R, Shekelle P.
Will this patient develop persistent disabling low back pain?
JAMA. 2010;303(13):1295-1302.
doi: 10.1001/jama.2010.344R.A. Deyo, S.F. Dworkin, D. Amtmann, G. Andersson, et al.,
Report of the NIH Task Force on Research Standards
for Chronic Low Back Pain
Journal of Pain 2014 (Jun); 15 (6): 569–585Hill JC, Whitehurst DG, Lewis M, et al..
Comparison of Stratified Primary Care Management For Low Back Pain
With Current Best Practice (STarT Back): A Randomised Controlled Trial
Lancet. 2011 (Oct 29); 378 (9802): 1560–1571Foster NE, Mullis R, Hill JC, et al.;
IMPaCT Back Study team . Effect of stratified care for low back pain
in family practice (IMPaCT Back): a prospective
population-based sequential comparison.
Ann Fam Med. 2014;12(2):102-111.
doi: 10.1370/afm.1625Hill JC, Dunn KM, Main CJ, Hay EM.
Subgrouping low back pain: a comparison of the STarT Back Tool
with the Orebro Musculoskeletal Pain Screening Questionnaire.
Eur J Pain. 2010;14(1):83-89.
doi: 10.1016/j.ejpain.2009.01.003Koes BW, van Tulder MW, Ostelo R, Kim Burton A, Waddell G.
Clinical Guidelines for the Management of Low Back Pain
in Primary Care: An International Comparison
Spine (Phila Pa 1976) 2001 (Nov 15); 26 (22): 2504–2513Koes BW, van Tulder M, Lin CW, Macedo LG, McAuley J, Maher C.
An Updated Overview of Clinical Guidelines for the Management of
Non-specific Low Back Pain in Primary Care
European Spine Journal 2010 (Dec); 19 (12): 2075–2094Oliveira CB, Maher CG, Pinto RZ, et al..
Clinical practice guidelines for the management of non-specific
low back pain in primary care: an updated overview.
Eur Spine J. 2018;27(11):2791-2803.
doi: 10.1007/s00586-018-5673-2Tucker HR, Scaff K, McCloud T, et al..
Harms and benefits of opioids for management of non-surgical
acute and chronic low back pain: a systematic review.
Br J Sports Med. 2020;54(11):664.
doi: 10.1136/bjsports-2018-099805Lemmers GPG, van Lankveld W, Westert GP, van der Wees PJ, Staal JB.
Imaging versus no imaging for low back pain: a systematic review,
measuring costs, healthcare utilization and absence from work.
Eur Spine J. 2019;28(5):937-950.
doi: 10.1007/s00586-019-05918-1Jacobs JC, Jarvik JG, Chou R, et al..
Observational study of the downstream consequences
of inappropriate MRI of the lumbar spine.
J Gen Intern Med. 2020;35(12):3605-3612.
doi: 10.1007/s11606-020-06181-7Mafi JN, McCarthy EP, Davis RB, Landon BE.
Worsening Trends in the Management and Treatment of Back Pain
JAMA Internal Medicine 2013 (Sep 23); 173 (17): 1573–1581Kamper SJ, Logan G, Copsey B, et –al.
What is Usual Care for Low Back Pain? A Systematic Review of
Health Care Provided to Patients with Low Back Pain in
Family Practice and Emergency Departments
Pain. 2020 (Apr); 161 (4): 694–702
There are many more articles like this on our
What is Usual Care? PageDelitto A, Patterson CG, Stevans JM, et al..
Study protocol for targeted interventions to prevent chronic low back
pain in high-risk patients: a multi-site pragmatic cluster
randomized controlled trial (TARGET Trial).
Contemp Clin Trials. 2019;82:66-76.
doi: 10.1016/j.cct.2019.05.010Hill JC, Dunn KM, Lewis M, et al.
A Primary Care Back Pain Screening Tool:
Identifying Patient Subgroups For Initial Treatment
(The STarT Back Screening Tool)
Arthritis and Rheumatism 2008 (May 15); 59 (5): 632–641Fairbank JC, Pynsent PB.
The Oswestry Disability Index
Spine (Phila Pa 1976) 2000 (Nov 15); 25 (22): 2940–2952Neighborhood Atlas.
Accessed January 11, 2021.
https://www.neighborhoodatlas.medicine.wisc.edu/mappingSeaman SR, White IR.
Review of inverse probability weighting for dealing with missing data.
Stat Methods Med Res. 2013;22(3):278-295.
doi: 10.1177/0962280210395740Atlas SJ.
Management of Low Back Pain: Getting From
Evidence-Based Recommendations to High-Value Care
Annals of Internal Medicine 2017 (Apr 4); 166 (7): 533-534Traeger AC, Buchbinder R, Elshaug AG, Croft PR, Maher CG.
Care for Low Back Pain: Can Health Systems Deliver?
Bulletin of the World Health Organization 2019 (Jun 1); 97 (6): 423–433George SZ, Goertz C, Hastings SN, Fritz JM.
Transforming Low Back Pain Care Delivery in the United States
Pain 2020 (Dec); 161 (12): 2667-2673Buchbinder R, van Tulder M, Öberg B, Costa LM, Woolf A, at al.
Low Back Pain: A Call For Action
Lancet. 2018 (Jun 9); 391 (10137): 2384–2388
This is the fourth of 4 articles in the remarkable Lancet Series on Low Back PainKorownyk C, McCormack J, Kolber MR, Garrison S, Allan GM.
Competing demands and opportunities in primary care.
Can Fam Physician. 2017;63(9):664-668Sowden G, Hill JC, Morso L, Louw Q, Foster NE.
Advancing practice for back pain through stratified care (STarT Back).
Braz J Phys Ther. 2018;22(4):255-264.
doi: 10.1016/j.bjpt.2018.06.003Cherkin D, Balderson B, Wellman R, et –al.
Effect of Low Back Pain Risk-stratification Strategy on
Patient Outcomes and Care Processes: The MATCH
Randomized Trial in Primary Care
J General Internal Medicine 2018 (Aug); 33 (8): 1324–1336Day CS, Yeh AC, Franko O, Ramirez M, Krupat E.
Musculoskeletal Medicine: An Assessment of the Attitudes
and Knowledge of Medical Students at Harvard Medical School
Academic Medicine 2007 (May); 82 (5): 452–457Finestone AS, Raveh A, Mirovsky Y, Lahad A, Milgrom C.
Orthopaedists’ and family practitioners’ knowledge of simple low back pain management.
Spine (Phila Pa 1976). 2009;34(15):1600-1603.
doi:10.1097/BRS.0b013e3181a96622Matzkin E, Smith EL, Freccero D, Richardson AB.
Adequacy of Education in Musculoskeletal Medicine
Journal of Bone and Joint Surgery 2005 (Feb); 87 (2): 310–314Allan GM, McCormack JP, Korownyk C, Lindblad AJ, Garrison S, Kolber MR.
The future of guidelines: primary care focused, patient oriented,
evidence based and simplified.
Maturitas. 2017;95:61-62. doi:10.1016/j.maturitas.2016.08.015Lau R, Stevenson F, Ong BN, et –al.
Achieving change in primary care—effectiveness of strategies
for improving implementation of complex interventions:
systematic review of reviews.
BMJ Open. 2015;5(12):e009993. doi:10.1136/bmjopen-2015-009993Murphy DR, Justice BD, Paskowski IC, Perle SM, Schneider MJ.
The Establishment of a Primary Spine Care Practitioner And Its Benefits
To Health Care Reform in the United States
Chiropractic & Manual Therapies 2011 (Jul 21); 19 (1): 17Fox J, Haig AJ, Todey B, Challa S.
The effect of required physiatrist consultation on surgery rates for back pain.
Spine (Phila Pa 1976). 2013;38(3):E178-E184.
doi:10.1097/BRS.0b013e31827bf40cStandaert CJ, Li JW, Glassman SJ, et –al.
Costs associated with the treatment of low back disorders:
a comparison of surgeons and physiatrists.
PM R. 2020;12(6):551-562. doi:10.1002/pmrj.12266van Harten WH
Turning teams and pathways into integrated practice units:
appearance characteristics and added value.
Int J Care Coord. 2018;21(4):113-116.
doi:10.1177/2053434518816529Draugalis JR, Plaza CM.
Best practices for survey research reports revisited:
implications of target population, probability sampling,
and response rate.
Am J Pharm Educ. 2009;73(8):142. doi:10.5688/aj7308142Morton SM, Bandara DK, Robinson EM, Carr PE.
In the 21st Century, what is an acceptable response rate?
Aust N Z J Public Health. 2012;36(2):106-108.
doi:10.1111/j.1753-6405.2012.00854.xGeorge SZ, Lentz TA, Beneciuk JM, Bhavsar NA, Mundt JM, Boissoneault J.
Framework for improving outcome prediction for acute to chronic low back pain transitions.
Pain Rep. 2020;5(2):e809. doi:10.1097/PR9.0000000000000809

Return to LOW BACK PAIN
Since 4-29-2023

| Home Page | Visit Our Sponsors | Become a Sponsor |
Please read our DISCLAIMER |