

The Etiologies, Pathophysiology,
and Alternative/Complementary
Treatment of AsthmaThis section was compiled by Frank M. Painter, D.C.
Send all comments or additions to: Frankp@chiro.org




FROM: Alternative Medicine Review 2001 (Feb); 6 (1): 20–47 ~ FULL TEXT
Alan L. Miller, NDIntroduction
Asthma is a chronic inflammatory disorder of the respiratory airways, characterized by increased mucus production and airway hyper-responsiveness resulting in decreased air flow, and marked by recurrent episodes of wheezing, coughing, and shortness of breath. It is a multifactorial disease process associated with genetic, allergic, environmental, infectious, emotional, and nutritional components. Because of their symptomatology the majority of individuals with asthma experience a significant number of missed work or school days. This can create a severe disruption in quality of life, often leading to depressive episodes. It also disrupts the lives of caregivers and family members of the affected individual. Asthma patients who have increased symptomatology at night (a significant portion) also tend to have disturbed sleep patterns and impaired daytime attention, concentration, and memory. [1]
In 1998 it was estimated that asthma affected 17.3 million individuals in the United States and 150 million worldwide. From 1980–1995 the incidence of asthma in children under age 18 increased five percent per year, resulting in an increase of more than 100 percent in that time period, according to the National Health Interview Survey (NHIS), the mechanism the U.S. government uses to gather data regarding asthma prevalence and mortality. The current overall prevalence in children is estimated at 6.0–7.5 percent, with a total of over five million children affected. Asthma is the fourth-leading cause of disability in children, and one of the most common reasons for school absenteeism. The prevalence in adults is approximately five percent. Asthma prevalence among African-Americans is considerably higher than Caucasians or Hispanics, with black children having a 26–percent greater incidence than white children in 1995–1996.
Approximately 5,000 people die each year due to asthma. Across racial and socioeconomic groups, the death rate from asthma mirrors the incidence, with African-Americans having the highest mortality from this disease. The death rates for asthma are higher in the inner city and in lower socioeconomic groups. The exact cause of these differences might be due to genetic, socioeconomic, and/or access to health care issues. Direct costs (doctors' visits, hospitalization, drugs, etc.) and indirect costs (work and school absenteeism, etc.) of asthma vary, depending on the reference, but are estimated to be approximately $6 billion per year.
Why the ever-increasing incidence of asthma in the last three decades? Some blame new home construction in the 1970s, when higher fuel costs prompted the construction of more airtight homes. Newer houses are more insulated and have less air exchange than older homes. Wall-to-wall carpet is much more common, as is central heating. Synthetic building materials laden with chemicals also enjoy greater utilization by builders. These "improvements" in construction make for a more closed micro-environment that has insufficient fresh air and is more conducive to the growth of microorganisms.
Other researchers point the finger at environmental pollutants. Industrialization of countries and the use of fossil fuels have paralleled the incidence of respiratory disease. There is good evidence that the increases in ozone, nitrogen dioxide, sulfur dioxide, and particulates in the atmosphere have exacerbated allergic diseases, including asthma, due to irritant effects of these substances causing chronic inflammation, as well as interactions with allergens and amplification of allergic reactions. [2, 3]
Changes in diet including an increased intake of omega-6 fatty acids and a decreased intake of nutrients such as magnesium and altered intestinal microflora are also hypothesized as contributors to the increased incidence of asthma. [2, 4, 5]
There is also the possibility that the practice of vaccinating children has contributed to this increase in asthma incidence, although presently this theory has not been studied thoroughly. Investigators in New Zealand, which has one of the highest rates of asthma in the world, found that 23 children who had not been immunized with the diptheria/tetanus/pertussis (DPT) and polio vaccines had no episodes of, or physician consultations for, asthma, whereas a group of immunized children had a 23-percent incidence of asthma. [6] Researchers in England note similar results in a survey of 446 children. In a group of 203 children who had not been immunized for pertussis, two percent had a diagnosis of asthma at eight years of age, compared to 11 percent of 243 who had been vaccinated for pertussis (p=0.0005). [7] However, Swedish researchers did not find this connection in a study of 9,000 children given either DPT or only the DT components. [8]
The Role of Inflammation in Asthma
The underlying patho-physiology of asthma, regardless of allergic components or triggering mechanisms, is airway inflammation. At the center of this improper inflammatory reaction is the T-cell. There is increasing evidence that the underlying process driving and maintaining the asthmatic inflammatory process is an abnormal or inadequately regulated CD4+ T-cell immune response to otherwise harmless environmental antigens. The major CD4+ T-cell subset involved in this process is the CD4+ Th2 subset, which produces a series of cytokines (secondary messaging molecules), including interleukin-4 (IL-4), IL-5, IL-6, IL-9, IL-10, and IL-13 (Table 1). These cytokines stimulate the growth, differentiation, and recruitment of mast cells, basophils, eosinophils, and B-cells, all of which are involved in humoral immunity and the allergic response. The other subset of CD4+ cells is the Th1 cell, which is responsible for production of interferon gamma (IFN-g) and interleukin-2 (IL-2), which are involved in delayed hypersensitivity responses and cellular immune responses to intracellular parasites and viruses. It is not yet known precisely why individuals with asthma have this overriding Th2 activity. It may be that genetics, viruses, fungi, heavy metals, nutrition, and pollution all contribute to this debilitating and sometimes deadly disease process (Table 2).
Antigen-specific IgE is partly responsible for initiation of an allergic response in asthma. Antigens cross-link with IgE on mast cells, which then spill their contents (histamine, leukotrienes) and further amplify the inflammatory response by damaging local tissue and attracting other lymphocytes. The regulation of IgE production involves interactions between antigen-presenting cells, and B and T lymphocytes. Antigen-presenting cells such as macrophages and dendritic cells present an antigen to CD4+ Th2 cells, which secrete cytokines that magnify the immune response. IL-4 produced by Th2 cells stimulates IgE production in B-cells, while IL-5 stimulates eosinophil differentiation and mobilization to inflammatory sites (Figure 1). IL-10 enhances the growth and differentiation of mast cells and very importantly inhibits the production of IFN-g. It appears the presence of excess IL-4 can also "switch" cytotoxic CD8+ cells from their normal production of IFN-g (which promotes antiviral and antitumor activity) to production of IL-4 and IL-5, further augmenting inflammatory activity. [9–18]
Table 1 Cytokines produced by
Th1 and Th2 cells

Table 2 Inducers of Th2 activity

Figure 1 Interactions of Th2 cells
with other cells
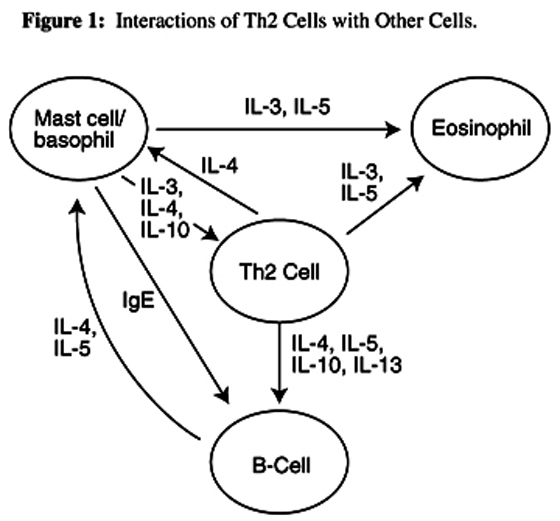
The inflammatory process is also promoted when histamine and leukotrienes are released by mast cells. Histamine acts very quickly and stimulates bronchoconstriction and excess mucus production. After the initial release of histamine, mast cells and other leukocytes manufacture and release leukotrienes, eicosanoid molecules that also enhance the inflammatory response. In this late-phase response, leukotrienes lipid-based molecules created by the action of the enzyme 5-lipoxygenase on arachidonic acid in cell membranes exacerbate the broncho-constriction brought on by histamine. Leukotriene B4 (LTB4) is a very potent mediator of bronchoconstriction and chemotaxis. The cysteinyl leukotrienes leukotrienes bound to the amino acid cysteine which include LTC4, LTD4, and LTE4, also attract leukocytes, in addition to their involvement in broncho-constriction and mucus production. The end result of these complex interactions is a cascading immune and inflammatory response characterized by airway eosinophilia, mucus hypersecretion, and airway hyper-responsiveness the hallmarks of asthma.
Etiologic Factors in Asthma
Allergies
Although asthma is a multifactorial condition, the strongest risk factor in the etiology of asthma is atopy (allergies, atopic dermatitis, allergic rhinitis). An atopic individual has a significantly greater probability of developing asthma, and persons with a family history of atopic disease are at greatest risk. It is accepted that an immunological response to various allergenic stimuli, including pet dander, dust mites, cockroaches, fungi, and foods is a major triggering factor in asthma symptomatology.
Estimates of the number of people with asthma who also have allergic rhinitis are as high as 80 percent. [19] Some practitioners suggest they are the same malady, only in different areas of the respiratory tract and should be treated similarly. [19] In one study, 79 percent of individuals with asthma also had chronic rhinosinusitis. [20]Dust mites: Indoor allergens are numerous; however, dust mites (Dermatophagoides pteronyssinus) contribute greatly to the overall antigenic load in asthmatic individuals. The average home provides optimal temperature, humidity, and other environmental conditions for dust mite growth and reproduction, although dust mites appear in greater numbers in warm, humid climates. Dust mite feces are the major antigenic component, containing at least 10 antigens. A number of epidemiological studies have shown a correlation between dust mite exposure and asthma symptoms. [21–24] A Swedish study found significantly greater risk for asthma symptomatology in homes with higher levels of dust mites. [25] A U.S. study of inner-city children with asthma discovered a high proportion of these children had both a significant exposure and an immunological sensitization to dust mites and cockroaches. [26]
Dust mite allergenicity could be at least partly due to enzymes in dust mite feces, which when inhaled can interrupt tight junctions between epithelial cells in the lungs. This disruption of the normal epithelial barrier can subsequently enhance the presentation of antigens (dust mite and others) to dendritic cells residing beneath the airway epithelium and facilitate immune response to those antigens. Such a mechanism would to some extent explain commonly-occurring allergenic responses to other inhalant antigens in individuals exposed and sensitized to dust mites. [27]
The allergic response to dust mites involves an immediate hypersensitivity response, including increases in specific IgE antibodies and T-cells of the Th2 phenotype. Inhalation challenge of the allergic individual with dust mite antigen produces airway hyper-reactivity and bronchospasm, along with an eosinophil-dominated inflammatory response.
Preventive hygienic measures to reduce dust mite exposure include washing bedding in hot water, eliminating carpet where possible, and encasing mattresses and pillows in occlusive covers. The use of high efficiency particulate air (HEPA) filters in heating, ventilation, and air conditioning systems is also helpful, as is the use of a vacuum cleaner with an on-board HEPA filter to clean floors and upholstery. [28]
Cockroaches: As with dust mites, cockroaches can be a very significant allergen source in the allergic asthmatic. It is thought that cockroach antigens (from the body and feces) pose a significant threat to individuals with asthma, and may be partially responsible for the greatly increased morbidity and mortality from asthma in inner-city residents. Results of The National Cooperative Inner-City Asthma Study (NCICAS) demonstrate the degree of exposure to cockroach antigen, measured in bedroom dust, is directly correlated with a sensitized child's risk of hospitalization. [29]
Dog and Cat Dander: Dogs and cats are another significant source of antigenic stimuli in the home, and can initiate or aggravate airway inflammation and asthma symptoms. [30] Cat dander tends to be more antigenic, as the allergen (which derives from salivary and sebaceous glands) is quite small, and thus can stay airborne for long periods of time. Conversely, dog allergen is larger and tends not to be airborne as much as cat allergen, making it more likely cat allergen will be inhaled. [31]
In a study involving 787 asthmatics, the presence of domestic animals in the home was one of the most significant predictors of asthma morbidity; [32] other studies have confirmed these results. [33–35] An interesting study was conducted in Los Alamos, New Mexico, which is at 7,200 feet elevation. At this altitude, household dust does not contain high levels of dust mite antigens. Among school children in this community with asthma, (n=57) sensitization to cat and dog allergens was strongly associated with bronchial reactivity and asthma symptoms, while mites, cockroaches, and pollens were not significantly associated with asthma symptoms. [36]
Unfortunately, strict avoidance of animal allergens is practically impossible, because even if domestic animals are not in the home there is still a possibility of significant exposure due to transfer of animal dander in public places. Studies of cat and dog allergens in Swedish schools discovered high concentrations of these allergens in school dust. [37, 38] Perzanowski et al found allergen levels higher than those found in homes without pets and noted, "The schools appear to be a major site of exposure to cat and dog allergens." [38] Significant levels of cat and dog allergens were also found in public waiting areas of a British hospital, although vacuuming three times per week decreased allergen levels in upholstered chairs. [39] Bathing animals can also be helpful in reducing the amount of allergen transfer. According to a recent British study, washing the pet dog twice per week can decrease airborne canine allergen 84 percent. [40]
It is vital to reduce total allergenic load, if possible. If a pet lives in the house, maximizing the time the animal spends outdoors is a must. Animals must not be allowed into the bedroom, which should be considered a "clean room" that has as few allergens as possible. This provides the asthmatic an environment that is as free as possible from allergens for at least eight hours out of 24. HEPA filters are a helpful adjunct in reducing the allergenic load from animal dander in the home. [41]
Food Allergy: Asthma can also be caused or exacerbated by food allergy. It is probably not as important a causative factor as inhalant allergies, but nevertheless contributes to the overall allergenic load of the asthmatic. Some estimates are that 5–8 percent of people with asthma have a food allergy that can be confirmed via a double-blind, placebo-controlled food challenge. [42–46] Patient estimates of food allergy in asthma are much higher, ranging from 20–60 percent. [45, 46] Many who make dietary modifications to avoid foods they believe are causing asthmatic symptoms feel those modifications help their asthma, including 79 percent of asthma patients in one study. [45]
Atopic individuals produce specific IgE antibodies to food proteins, which bind primarily to mast cells and basophils. When the person comes in contact with the allergic food, histamine is released by these cells, stimulating an inflammatory response in the lungs, and asthma symptoms ensue. This reaction, or the involvement of non-IgE-mediated reactions, can be immediate or may take hours or days to manifest. This might be one reason why estimates of food allergy in asthma patients are low, as researchers often look for symptoms to appear quickly following challenge; whereas, with a delayed reaction to foods, it is more difficult to assess the cause of the symptoms.
Gastrointestinal symptoms occur more frequently in children with asthma and atopic dermatitis; [47] and abnormal gastrointestinal permeability is found in a greater percentage of asthmatics compared to non-asthmatic controls. [48] It is possible there is a common defect in the respiratory and gastrointestinal mucosa, either caused by the asthma or as a possible cause of asthma. Increased gastrointestinal permeability can allow large antigenic molecules to be absorbed through the mucosa, causing sensitization to foods. Possibly the increased permeability in the lungs caused by dust mite antigen causes a similar increase in transfer of antigenic material across the respiratory epithelium. [27]
Although the role of food allergies as a causative factor in asthma remains ambiguous, it seems some individuals do benefit from avoidance of identified problem foods. This option should not be overlooked in dealing with asthma.
Yeast/Fungi: Fungi are known to be causative factors that induce asthmatic symptoms. Outdoor airborne fungi, including Cladosporium, Alternaria, Penicillium, and Aspergillus are significant triggers of IgE formation, as are the indoor fungi Aspergillus, Neurospora, and Eurotium. In addition, some practitioners believe there is a strong fungal/yeast component in the lung and/or gut microflora in individuals with asthma. [31, 49, 50] Ridding the home or work environment of these organisms and utilizing antifungal treatments as appropriate has been reported to improve asthma symptomatology.
Subjective symptoms and peak expiratory flow (PEF) in 74 children with asthma were followed for 16 weeks, and correlated with the amount of bacterial endotoxin and fungal 1,3-beta glucan levels present in house dust. After adjusting for pet presence, type of floor cover, and dust mite allergen levels, yeast levels were positively correlated with PEF variability in these children. [51]
Sensitivity to fungal allergens has also been found to be a risk factor for severe life-threatening asthma. A New Zealand study of patients admitted to a hospital intensive care unit (ICU) revealed that patients admitted to the ICU had a significantly greater incidence of reactivity to Alternaria tenuis, Cladosporium cladosporoides, Helminthosporium maydis, or Epicoccum nigrum (54% vs 30% for other groups not admitted to the ICU or not hospitalized for asthma). [52]
Fungal cultures were performed from bronchial secretions of 13 asthma patients and from the skin of 91 patients with atopic dermatitis. The predominant yeast species present on the skin were Candida and Rhodotorula species, while Candida species were the most prominent species isolated from bronchial secretions. [53] Candida albicans may well be a prominent allergen for people with asthma. The cell wall constituent, mannan, and acid protease an enzyme secreted by C. albicans are both highly allergenic, and serum IgE antibodies are often increased in atopic individuals. [54, 55]
Animal and in vitro studies suggest if there is an imbalanced Th1/Th2 ratio of immune activity, Candida infection is more likely to occur. [56–60] There has been little investigation to date as to whether asthmatics are more likely to have Candida infections because of Th2-dominance, or whether Candida infection predisposes an individual to experience asthma symptoms. What data there is suggests both may be true. [49, 50, 61, 62] Regardless, these data suggest that environmental fungi and/or colonization with Candida or other organisms probably contribute to asthma severity. Environmental eradication of fungi, as well as internal antifungal agents, should be considered in those testing positive for reactivity to these organisms.
A Probable Connection to Persistent Viruses and Chlamydia
The search for etiologic agents in bronchial asthma has brought some researchers to believe viruses or other organisms might be partly responsible for some asthma cases. Infections with common cold viruses and influenza frequently precipitate symptoms in those with established asthma. The chief mechanism for these exacerbations appears to be viral replication in respiratory epithelial cells triggering cytokine release, inflammation, and mucus production. These processes are necessary to clear the viral infection; but superimposed over a pre-existing inflammatory condition in the airways they can trigger symptomatology.
There is evidence that some viruses may, in the presence of IL-4 (produced in excess by Th2 cells in asthmatics) cause CD8+ cells to drastically reduce their usual secretion of IFN-g and switch to production of IL-4 and IL-5. This switch to greater Th2 activity can slow the immune system's clearance of the virus, cause pulmonary eosinophil infiltration, and exacerbate asthma inflammation. [18] Marin et al reported that 39 of 50 (78%) presently asymptomatic asthmatic children harbored adenovirus DNA in their nasopharynx, detected from nasal swabs, while the virus was found in only one of 20 controls (5%). Rhinovirus was found in 16 asthma patients (32%), and none of the controls. The authors believe the presence of these viruses in patients, but not controls, might reveal a possible connection between viral infections and asthma. [63]
It also has been hypothesized that Chlamydia pneumoniae infection might predispose individuals to asthma. A recent review on this subject examined papers over a 15-year period; epidemiological studies and case reports were included. The authors found significant epidemiological associations between Chlamydia infection and asthma, as well as reports of clinical improvement following antibiotic treatment for this organism. [64] A study of children and young adults with asthma found the opposite higher IgG titers for Chlamydia were correlated with a reduced risk of asthma. [65] Another study related high IgG and IgA titers with lower FEV1 (Forced Expiratory Volume in One Second an objective measurement of airflow); i.e., higher titers of antibodies to Chlamydia were associated with increasing severity of asthma in adults. [66] There is possibly a difference in how Chlamydia might affect children compared to adults, which might explain the disparity in these results.
Another virus with a possible link to asthma symptoms is respiratory syncytial virus (RSV). Wheezing is a cardinal sign of acute RSV infection in infancy, and may persist chronically for years after an acute infection. In a 10-year follow-up of children hospitalized due to RSV infection, 40 percent reported wheezing at five years (11 percent in controls), and 22 percent reported wheezing at 10 years (10 percent in controls). [67] A Brazilian study confirms these findings, demonstrating a significantly increased risk for wheezing at age six in children who had RSV before age three. The risk for wheezing decreased with increasing age, becoming insignificant by age 13. [68] These studies show that, while RSV infection in a young child is a significant risk factor for wheezing until ages 10–13, it does not pose a significant risk for asthma symptoms in adults. It has been proposed that RSV infection stimulates an over-active Th2 cytokine response [69, 70] which, as noted above, predisposes toward eosinophilia and hyper-reactive airways (Table 2).
Indoor and Outdoor Air Pollution
An increase in inhaled particulate matter, whether from cigarette smoking, environmental tobacco smoke, fossil fuels, or wood burning stoves can exacerbate asthma symptoms. Recent animal research reveals second-hand smoke up-regulates the Th2 immune response. This mechanism might partially explain epidemiological evidence linking second-hand smoke with asthma prevalence. [71] Use of gas appliances in the home can increase the concentration of nitrogen dioxide in inspired air, correspondingly reducing lung function. Volatile organic compounds and formaldehyde in the indoor environment, from off-gassing of paints, adhesives, furnishings, and building materials can also increase the risk of asthma attacks. An excellent review of the subject of asthma and the home environment can be found in Jones' recent article in the Journal of Asthma. [31]
Does Heavy Metal Toxicity Promote Inflammation in Asthma?
Lead and mercury toxicity has been shown in animal studies to inhibit Th1 cells and stimulate a Th2 immune response and, in some animals, promote autoimmune diseases. [11, 72] In humans, if heavy metals were proven to cause an imbalance in the normal Th1:Th2 ratio, it would explain practitioner reports of improvements in asthma symptoms after heavy metal detoxification.
Gastroesophageal Reflux: a Possible Connection
Another possible contributor to the etiology of asthma is gastroesophageal reflux (GER). An increased incidence of GER has been noted in asthma patients; [73, 74] however, it is not fully understood if these conditions simply overlap, if GER causes or exacerbates asthma, or if asthma causes GER. In his 2000 review, Sontag [74] estimates that approximately 75 percent of asthmatic patients experience GER symptoms, 80 percent have abnormal acid reflux, 60 percent have a hiatal hernia, and 40 percent have esophageal damage (erosions or ulcerations). Two mechanisms have been proposed to explain how GER might cause asthma symptoms:(1) a vagal-mediated reflex from the irritated esophagus to the lung, causing reflex bronchoconstriction, or
(2) microaspiration of gastric acid, causing pulmonary irritation, injury, and subsequent overproduction of mucus.Proponents of the vagal reflex theory note that the esophagus and lungs share the same embryonic tissue and innervation. An irritated esophagus could, therefore, initiate a vagal reflex, resulting in increased bronchial reactivity. [74–79] Animal and human studies have shown increased pulmonary airway resistance with esophageal acid infusion. This bronchoconstriction was reversed with antacid therapy [80] or surgical interruption of the vagus nerves. [74] In a small study of pediatric asthma (n=9), acid was infused into the esophagus during sleep, causing bronchoconstriction only in the four children with previously diagnosed esophagitis (positive Bernstein test). These results led the researchers to comment that reflux, an irritated esophagus, and a low nocturnal threshold to bronchoconstrictive stimuli were all necessary for reflux to cause bronchoconstriction. It is interesting to note that, in this study, acid did not cause bronchoconstriction during a midnight infusion, but did in the 4–5 a.m. infusion. [78] In a study of 47 adults (20 asthmatics with reflux, 7 without, 10 participants with GER, and 10 controls) esophageal acid infusion caused a significant decrease in PEF in all participants; however, the group of asthma patients with reflux had a greater decrease in PEF, as well as further deterioration after the acid was cleared with normal saline. The authors stated that since there was no microaspiration of acid, a vagally mediated reflex must be involved. [77] Subsequent studies found increased vagal responsiveness in patients with asthma and GER; this was blocked by atropine, which inhibits vagal stimulation. [81, 82]
Animal and human studies have provided evidence for the microaspiration theory of GER, although overall the evidence looks less convincing than the "reflex theory." Inhalation of a dilute acid solution in cats caused significantly greater bronchoconstriction than infusion of acid into the esophagus. [83] Ambulatory esophageal pH monitoring and scintigraphic technetium monitoring have provided documentation of esophageal acid reflux and microaspiration of gastric acid in humans. [84, 85] It might be that asthma related to GER is a multifactorial problem, with components of microaspiration and gastroesophageal reflux.
If indeed GER causes asthma or exacerbates hypersensitive respiratory tissue, the true test should be that anti-reflux or antacid therapy significantly improves asthma symptoms. However, antacid therapy, which has consisted mostly of H2 blockers (cimetidine, ranitidine) or a proton pump inhibitor (omeprazole), has not been consistently effective, showing mixed results. [74]
A small study (n=5) of asthma patients with nocturnal symptoms and GER determined that treating asthma with ephedrine improved asthma symptoms as well as reflux symptoms. The authors stated the bronchodilation provided by the ephedrine and the subsequent improvement in GER symptoms suggests GER might be a result of asthma symptomatology, not the opposite. [86]
The most common GER therapy is a pharmacological reduction in gastric acid output. However, Wright found a substantial number of children with asthma actually have a reduction in gastric acid output. He theorizes that reduced gastric output results in inadequate protein digestion and an increase in allergenicity of foods, as well as a reduction in nutrient absorption. Treatment with hydrochloric acid supplementation is part of his integrated treatment protocol, and is claimed to provide symptomatic improvement. [87, 88]
The Possible Role of Dehydration in Asthma
It is important to ensure the asthma patient is well hydrated; however, good data does not exist showing a firm association between dehydration and asthma, except in exercise-induced asthma (EIA). In EIA, dehydration of airway epithelial cells may contribute to epithelial damage, edema, and hyper-responsiveness. [89–93] This does not rule out the possibility that chronic sub-clinical dehydration may contribute to asthma-related symptoms; in fact, it lends credibility to the theory.
Aspirin-induced Asthmatic Exacerbation
A subset of individuals with asthma experience symptoms after ingestion of aspirin or other similar non-steroidal anti-inflammatory drugs (NSAIDs). Since most NSAIDs block the enzyme cyclooxygenase, it is thought this leaves more arachidonic acid to react with the other arm of the eicosanoid pathway, regulated by activity of lipoxygenase. Downstream metabolites of this pathway include the leukotrienes, very potent stimulators of inflammation and bronchial constriction. Avoidance of NSAIDs is imperative in these individuals. Some asthmatics also react to sulfites present in some foods and wines. This reaction is more common in people who experience symptoms following ingestion of NSAIDs. [95]
The Emotional Connection
It is often said that asthma can be triggered by emotional stress. In fact, traditional Chinese medicine refers to the lungs in connection with grief and sorrow. Asthma patients have been noted to have a more negative affect, and emotional upheavals have been linked to asthma symptom exacerbations. In a recent study, Cetanni et al [95] examined 80 patients with asthma, 40 patients with either hepatitis B or C, and 40 healthy controls. Significantly greater anxiety and depression were found in asthma patients compared to hepatitis patients and controls. In a study of 230 patients with asthma, 45 percent scored high enough on depression ratings scales to be considered depressed. Those with more depressive symptoms reported worse health-related quality of life than asthma patients without depression. [96] This begs the question, do a significant number of asthma patients have anxiety and depression because of their asthma, or do these psychological diagnoses predispose one to asthma symptoms? It may be a combination of both. No doubt it is an anxiety-producing feeling when one cannot get enough air. Conversely, intense emotions can bring about asthma symptoms. Increased respiratory resistance, airway reactivity, shortness of breath, and decreased peak expiratory flow rate have been reported after an emotional challenge. [97–99]
Nutrients and AsthmaReferencesVitamin C There is reason to believe oxygen radicals are involved in the pathophysiology of bronchial asthma. Inflammatory cells generate and release reactive oxygen species, [100] and inflammatory cells from asthma patients produce more reactive oxygen species than non-asthmatics. [101, 102] Significantly decreased levels of vitamin C and vitamin E were found in lung lining fluid of asthmatics in a recent study, even though plasma levels were normal. [103] Fourteen children with asthma were found to have significantly decreased serum levels of vitamin E, beta-carotene, and ascorbic acid during an asymptomatic period, with elevated levels of lipid peroxidation products during an asthma attack. [104] To combat the increased oxidant burden in asthmatics, the attainment and maintenance of optimal levels of antioxidant nutrients might be essential.
Epidemiological studies of vitamin C intake and asthma symptoms and respiratory function note a beneficial overall effect of vitamin C. Generally, as vitamin C intake rises, FEV1 and FVC (forced vital capacity) increase. [105, 108] Yet the effect of vitamin C on asthma remains controversial, as studies on vitamin C supplementation in asthma patients have yielded contradictory results. For example, asthma patients subjected to methacholine challenge testing alone and after ascorbic acid supplementation (1 g one hour prior to challenge) were able to withstand greater doses of methacholine after vitamin C dosing. [109] In this test, methacholine, a bronchoconstricting drug, is inhaled. In those with hyper-reactive airways, there will be a greater constriction of pulmonary smooth muscle and loss of lung function. However, short-term dosing with ascorbic acid failed to improve bronchial hyper-reactivity with inhaled histamine challenge. [110] Schachter and Schlesinger studied the effect of ascorbic acid on exercise-induced asthma, and concluded that ascorbic acid has a mild bronchodilatory effect in exercise-induced bronchospasm, seen as a protective effect on FEV1 and FVC compared to placebo. [111]
Reviews regarding vitamin C and asthma point to the fact that the studies performed to date, whether showing positive or negative effects, utilized short-term vitamin C dosing, as if they were attempting to assess an immediate effect of vitamin C only, and not the effects of long-term optimal blood and tissue levels of this nutrient. [112, 113] Long-term supplementation studies of vitamin C, asthma symptomatology, and pulmonary function need to be conducted to further elucidate vitamin C's role in asthma treatment.
Vitamin B6 Pyridoxal 5'-phosphate (PLP), the active form of vitamin B6 in the body, is involved in numerous biochemical processes, and has been found in lower concentrations in asthma patients. [114] However, investigations of the therapeutic efficacy of B6 supplementation have resulted in mixed results. Treatment of asthma with pyridoxine (50 mg twice daily) resulted in improvements in a reduction of asthma exacerbations and wheezing episodes in adults. [114] In 76 children with asthma, B6 supplementation (100 mg pyridoxine HCl twice daily) resulted in fewer bronchoconstrictive attacks; less wheezing, cough, and chest tightness; and less use of bronchodilators and steroid medications. [115] A double-blind trial of B6 (300 mg/day pyridoxine HCl) in steroid-dependent asthma patients resulted in no change in lung function. [116]
Asthma patients treated with the bronchodilator theophylline have lower blood levels of PLP, possibly due to PLP depletion secondary to its use in theophylline metabolism. Theophylline is not used as much as it once was, mostly due to side effects and its narrow therapeutic range; [117, 118 ] however, monitoring of vitamin B6 levels and supplementation if warranted should be considered for individuals using this drug.
Vitamin B12 It has been reported that children with asthma may be B12 deficient, although there is no peer-reviewed literature to corroborate such a statement. Jonathan Wright, MD, and Alan Gaby, MD, relate that asthmatic children respond well to B12 supplementation, particularly if they are sulfite-sensitive. Daily doses of 1000–3000 mcg may be needed. [119]
Magnesium Magnesiumis a cofactor in over 300 biochemical processes in the body, and is especially vital to the contraction/relaxation state of smooth muscle. Magnesium and calcium work in concert to regulate the contraction and relaxation of smooth muscle. Low magnesium enhances the contraction activity of calcium, while higher magnesium levels inhibit calcium and promote relaxation. Hypomagnesemia is common in asthmatics, [120–123] and worsens in more severe cases. [120, 121]
Serum levels are often used to assess magnesium status; however, serum magnesium can be normal while intracellular magnesium is deficient. Intracellular assessment utilizing erythrocytes or leukocytes is recommended for an accurate depiction of magnesium status. Intracellular magnesium was assessed in 22 asthma patients and compared with 38 controls with allergic rhinitis. Magnesium levels were significantly lower in individuals with asthma versus controls. Lower intracellular magnesium was correlated with increased airway hyper-reactivity via the methacholine challenge test. Magnesium levels did not significantly affect FEV1. [122] Similar findings were recently reported by Hashimoto et al. [121] While low magnesium status is a consistent finding, the role of magnesium supplementation is more ambiguous.
A large British study of dietary magnesium intake and asthma symptoms in 2,633 people found individuals who had a greater dietary intake of magnesium had a significantly higher FEV1 and significantly decreased airway hyper-reactivity. [124] In a randomized, double-blind, placebo-controlled crossover study, Hill et al reported significantly fewer asthma symptoms and reduced subjective bronchial hyper-reactivity in patients given 400 mg magnesium per day as a dietary supplement. However, objective measurements of pulmonary function were not significantly better in the three-week study, and use of short-acting beta agonist inhaler medications was not decreased. [125] It might be that a three-week trial, while seeming to improve aspects of patient subjective symptomatology, is not long enough to have a long-term stabilizing effect on pulmonary function. An investigation on the effects of long-term magnesium supplementation to correct tissue levels of this mineral seems warranted.
Intravenous magnesium sulfate is a critical treatment component for severe asthma seen in the emergency department in many hospitals. Intravenous magnesium often relieves symptoms soon after infusion is begun, [126] and can decrease the need for intubation in status asthmaticus [127] and respiratory failure. [128] In recent pediatric studies, addition of magnesium sulfate IV to standard emergency care initiated faster improvement in PEF and oxygen saturation in patients not responsive to conventional treatments. [129, 130]
Another pediatric study of 30 patients with an acute asthma exacerbation used a high-dose protocol of 40 mg/kg magnesium sulfate infused over a 20-minute period. Significant improvement was noted at 20 minutes, and a much greater improvement was noted at 110 minutes: PEF had improved 26 percent (vs. 2% in saline controls), FEV1 24 percent (2%), and FVC 27 percent (3%). All results were highly significant (p<0.001). [131]
Acute administration of intravenous magnesium has been studied as a stand-alone therapy, as well as an adjuvant to conventional beta-adrenergic, methyl xanthine, and steroid treatment. Results have been mixed, with some studies finding statistically significant improvements in lung function [130–134] and others determining that IV magnesium sulfate is not helpful. [135–136] In two recent reviews of the subject, IV magnesium sulfate was found in one review to be of significant benefit to patients with severe asthma, [137] and found not to affect treatment outcomes in a meta-analysis. [138]
It is unknown why these various investigations resulted in diverse outcomes. Some intravenous trials used 1–2 g magnesium sulfate alone, while others used a similar dose as an initial bolus, followed by slower drips over the next few hours. There does not seem to be a pattern of results following a specific type of protocol. Regardless of the inconsistent results seen with IV magnesium sulfate, asthmatics do tend to have lower intracellular levels of magnesium, and supplementation to correct those levels seems warranted.
Zinc There is little direct evidence of zinc deficiency causing asthma symptoms, but asthma patients have been shown to have lower plasma zinc than healthy controls. [139] Serum and hair zinc were significantly lower in individuals with asthma and atopic dermatitis. [140] Similar results were reported by Di Toro et al in asthmatics versus controls. [141]
It has been proposed that a zinc deficiency switches the Th1 immune response toward a Th2-type response, which, as mentioned earlier, is a hallmark of asthma pathophysiology. [142,143] Prasad et al studied how mild zinc deficiency affects the immune system. Zinc deficiency caused an imbalance between Th1 and Th2 functions, with a subsequent increased production of IL-4, IL-6, and IL-10, and decreased production of IL-2, IFN-g, and tumor necrosis factor alpha. They also noted decreased NK-cell activity and decreased numbers of cytotoxic CD8+ T-cell precursor cells. [144] In two other studies of zinc and immunity, individuals deficient in zinc exhibited diminished Th1 activity, but unaffected Th2 activity, creating a relative Th1 deficiency. [145, 146]
Even without the benefit of definitive research on long-term zinc supplementation in asthma patients, this author believes it is vitally important to ensure proper zinc nutriture in asthma patients to avoid a potential zinc-deficiency-induced exacerbation of asthma symptoms due to increased Th2 immune activity.
Selenium Glutathione is a vital component of the body's antioxidant system. Glutathione peroxidase (GSH-Px) is the selenium-containing enzyme that uses glutathione as a cofactor to metabolize hydrogen peroxide, and can thus protect against oxidative damage. Individuals with asthma tend to have increased oxidative activity, lowered selenium status, and decreased activity of glutathione peroxidase. [147–150]
Only one study has been conducted on selenium supplementation to combat the increased pulmonary oxidative burden in asthmatics. Hasselmark et al performed a double-blind, placebo-controlled study in which asthma patients were given 100 mcg sodium selenite (containing 46 mcg elemental selenium) for 14 weeks. Significant increases were seen in serum and platelet selenium and GSH-Px activity, and improvements were seen in subjective symptomatology. However, objective measurements of lung function were not changed. [151] It may be that the supplemental dosage given was too low and that a supplemental dose of 200–250 mcg might be more beneficial. More study is warranted in this important arena.
Omega-3 Fatty Acids Intermediate and end-products of fatty acid metabolism are known to have potent effects on the inflammatory process. Prostaglandins and leukotrienes from arachidonic acid metabolism are highly inflammatory molecules, and play an important role in the pathophysiology of asthma. Arachidonic acid is released from cell membrane phospholipids of activated immune cells (via activity of the enzyme phospholipase A2) in response to various immunological stimuli. Prostaglandins and leukotrienes resulting from arachidonic acid metabolism are pro-inflammatory molecules. Leukotriene B4 (LTB4) is involved in bronchoconstriction and leukocyte chemotaxis, while the cysteinyl leukotrienes LTC4, LTD4, and LTE4 are far more potent promotors of smooth muscle constriction and mucus production. An overabundance of these leukotrienes is implicated in the pathophysiology of asthma.
Research into the effects of leukotrienes has spurred the development of new drugs that block the activity of these potent substances. These drugs appear to be of benefit in some asthma patients, particularly those with more severe disease. Steroid medications, either inhaled or systemic via oral or parenteral dosing, have been the mainstay of anti-inflammatory asthma drug therapy. In contrast to the new leukotriene-inhibiting drugs, corticosteroids strongly inhibit the release of arachidonic acid from cell membranes by blocking the activity of phospholipase A2, resulting in a greatly diminished amount of prostaglandins and leukotrienes. Two LTD4 receptor antagonists, zafirlukast and montelukast, have been approved for use in asthma and provide moderate improvements in objective lung function tests, as well as less reliance on inhaled steroid medications. A 5-lipoxygenase inhibitor, zileuton, has demonstrated similar results. [152, 153] Cromolyn sodium, used prophylactically in asthma, has been shown to inhibit LTE4-induced bronchoconstriction, probably by inhibiting mast cell degranulation. [154]
Cold-water fatty fish contain relatively large amounts of the omega-3 fatty acids eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA). When these fish are eaten, or when oil derived from them is taken as a supplement, EPA and DHA displace arachidonic acid from cell membranes. When these cells are stimulated they subsequently release relatively higher concentrations of fish-derived oils. The resultant downline metabolites of EPA and DHA differ from arachidonic acid metabolites. EPA and DHA are converted by cyclooxygenase into 3-series prostaglandins, and by lipoxygenase to 5-series leukotrienes, both categories of which are far less potent inflammatory mediators than the 2-series prostaglandins and 4-series leukotrienes arising from arachidonic acid metabolism [155, 156] (Figure 2). Because of this shift toward less inflammatory eicosanoids, one would expect to see less inflammatory activity in the lungs, and a subsequent improvement in asthma symptoms and lung function. Epidemiological studies of dietary fish intake and risk of asthma show an inverse correlation; i.e., more fish consumed equals less risk of asthma. [157, 158] However, the clinical data is equivocal, with well-designed studies showing both positive and negative results from omega-3 fatty acid supplementation.
Figure 2 Fatty acids and inflammation
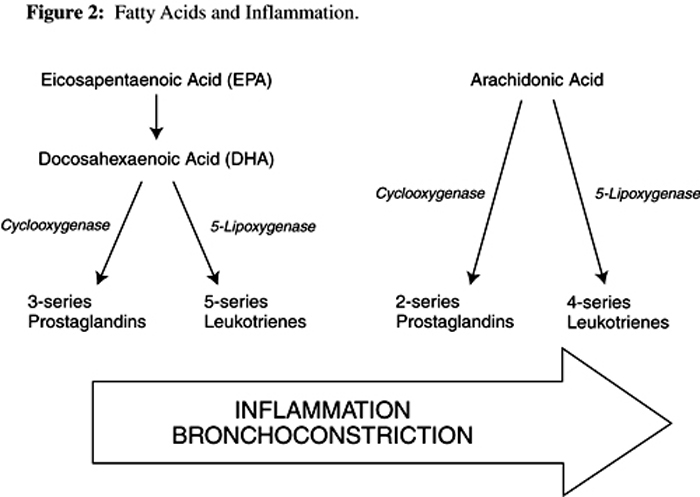
Click to enlarge
A group (n=7) of individuals with seasonal asthma were supplemented with 3 g/d of a fish oil concentrate containing approximately 1,300 mg each of EPA and DHA, resulting in decreased residual volume (which is usually increased in asthma patients) and decreased bronchial reactivity. [159]
Broughton et al studied 26 asthma patients after a one-month regimen of low- or high-dose omega-3 fatty acid intake. Patients' dietary intake of fish was evaluated, then supplementation was individualized for each patient so they ingested omega-3 and omega-6 fatty acids in a ratio of 0.1:1 or 0.5:1. This provided either 0.7 grams EPA/DHA or 3.3 grams EPA/DHA, respectively (the ratio of EPA to DHA in this study was not given). The high-dose protocol stimulated an improvement in bronchial reaction to methacholine challenge in 40 percent of subjects, compared to a reduction in lung function in the low-dose group. Leukotriene B5 was increased in the high-dose group and was predictive of lung function. [160] This seems to indicate a role for fish oil supplementation in asthma treatment.
In a separate study, after 10 weeks of supplementation with 3.2 g EPA and 2.2 g DHA per day, 12 subjects underwent histamine challenge, exercise challenge, and neutrophil studies to assess the efficacy of fish supplementation in asthma. Although there was a significant increase in omega-3 fatty acid content of neutrophils and a 50-percent inhibition of LTB4 synthesis, there was no detectable change in the clinical outcome; e.g., no significant change in histamine response, exercise response, FEV1, or symptom score. [161] Hodge et al reported similar results in their study of asthmatic children. After six months' supplementation with 1.2 g/d of omega-3 fatty acids, a five-fold increase in plasma EPA and a decrease in peripheral blood eosinophils was seen, but there was no change in symptom severity. [158] The reason for these mixed findings is not known.

Return to ASTHMA
Since 2–01–2001


| Home Page | Visit Our Sponsors | Become a Sponsor |
Please read our DISCLAIMER |