Observed Patterns of Cervical Radiculopathy:
How Often Do They Differ From a Standard,
"Netter Diagram" Distribution?This section is compiled by Frank M. Painter, D.C.
Send all comments or additions to: Frankp@chiro.org




FROM: Spine J. 2019 (Jul); 19 (7): 1137–1142 ~ FULL TEXT
Steven J. McAnany MD , John M. Rhee MD , Evan O. Baird MD , Weilong Shi MD , Jeffrey Konopka MD , Thomas M. Neustein MD , Rafael Arceo MD
Department of Orthopedic Surgery,
Hospital for Special Surgery,
535 East 70th St,
New York, NY 10021, USA.BACKGROUND CONTEXT: Traditionally, cervical radiculopathy is thought to present with symptoms and signs in a standard, textbook, reproducible pattern as seen in a "Netter diagram." To date, no study has directly examined cervical radicular patterns attributable to single level pathology in patients undergoing ACDF.
PURPOSE: The purpose of this study is to examine cervical radiculopathy patterns in a surgical population and determine how often patients present with the standard textbook (ie, Netter diagram) versus nonstandard patterns.
STUDY DESIGN/SETTING: A retrospective study.
PATIENT SAMPLE: Patients who had single-level radiculopathy with at least 75% improvement of preoperative symptoms following ACDF were included.
OUTCOME MEASURES: Epidemiologic variables were collected including age, sex, weight, body mass index, laterality of symptoms, duration of symptoms prior to operative intervention, and the presence of diabetes mellitus. The observed pattern of radiculopathy at presentation, including associated neck, shoulder, upper arm, forearm, and hand pain and/or numbness, was determined from chart review and patient-derived pain diagrams.
METHODS: We identified all patients with single level cervical radiculopathy operated on between March 2011 and March 2016 by six surgeons. The observed pattern of radiculopathy was compared to a standard textbook pattern of radiculopathy that strictly adheres to a dermatomal map Fisher exact test was used to analyze categorical data and Student t test was used for continuous variables. A one-way ANOVA was used to determine differences in the observed versus expected radicular pattern. A logistic regression model assessed the effect of demographic variables on presentation with a nonstandard radicular pattern.
RESULTS: Overall, 239 cervical levels were identified. The observed pattern of pain and numbness followed the standard pattern in only 54% (129 of 239; p=.35). When a nonstandard radicular pattern was present, it differed by 1.68 dermatomal levels from the standard (p<.0001). Neck pain on the radiculopathy side was the most prevalent symptom; it was found in 81% (193 of 239) of patients and did not differ by cervical level (p=.72). In a logistic regression model, none of the demographic variables of interest were found to significantly impact the likelihood of presenting with a nonstandard radicular pattern.
CONCLUSIONS: Observed patterns of cervical radiculopathy only followed the standard pattern in 54% of patients and did not differ by the cervical level involved. Cervical radiculopathy often presents with a nonstandard pattern. Surgeons should think broadly when identifying causative levels because they frequently may not adhere to textbook descriptions in actual clinical practice. We observed III level of evidence.
KEYWORDS: ACDF; Anterior cervical discectomy and fusion; Cervical radiculopathy; Cervical spine; Netter diagram; Pain pattern
From the Full-Text Article:
Introduction
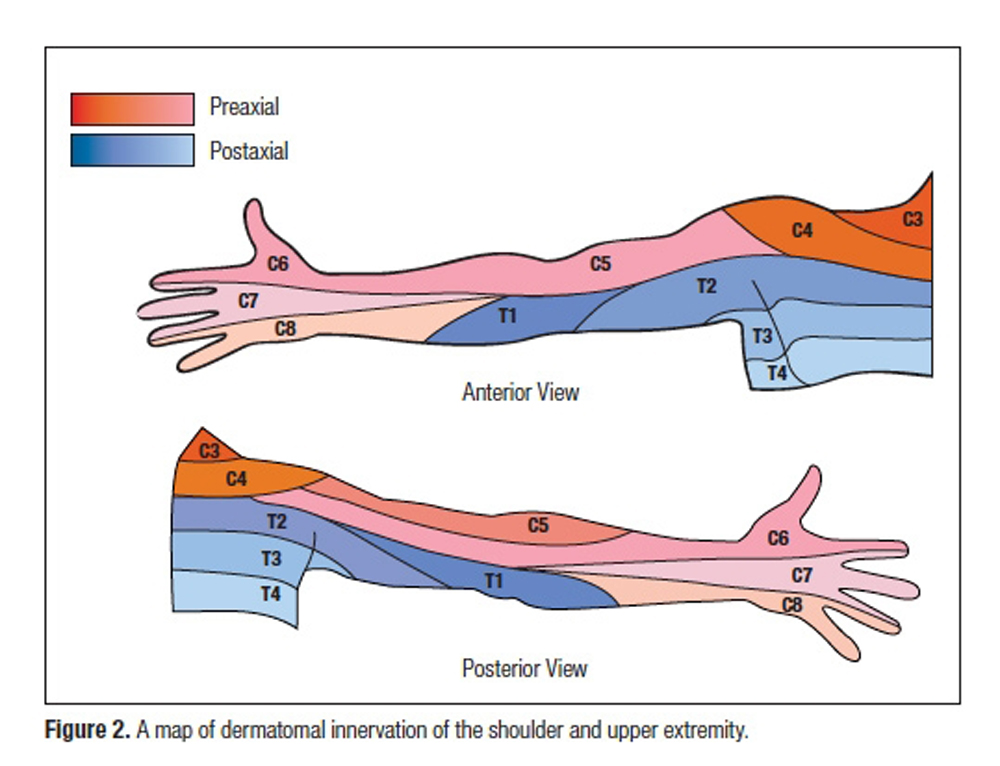
Traditionally, it is taught that cervical radiculopathy presents in a reproducible dermatomal and myotomal pattern attributable to the involved cervical root level. [1–3] Thus, for example, a C6 radiculopathy is classically described as symptoms radiating from the neck down the arm, terminating in the thumb and index finger. If advanced imaging shows root compression consistent with the clinically suspected level, a specific diagnosis can be made. Identification of the exact root level(s) causing radiculopathy can be important in all patients but is absolutely critical in those who elect to have surgery for refractory symptoms.
In clinical practice, however, patients may present with radicular symptoms that do not perfectly fit the standard, textbook “Netter diagram” patterns based on the description by Keegan and Garrett [13] Under these circumstances, proper identification of levels to target surgically can be difficult in those who fail nonoperative care. Uncertainty can lead to unnecessarily treating a level that is not actually causing symptoms or no surgery at all in a patient who might benefit from it for fear that the proper symptom generator can not been identified.
Brachial plexus variability is one potential cause of symptom variability in cervical radiculopathy. Anatomic dissections suggest that a typical “textbook” brachial plexus is only observed in 37–77%. [4–6] Additionally, a number of anatomic studies have demonstrated considerable variation in the intradural connection of rootlets, which can potentially impact the ability to accurately localize the level of cervical pathology on the basis of clinical signs and symptoms. [7–11] While the anatomic variability makes a compelling rationale for clinical symptom variability, clinically-based assessments of cervical radicular patterns remain limited. One study reported more than two-thirds (69.7%) of cervical radiculopathy patients exhibited non-dermatomal radiculopathy symptoms. [12] While this data is intriguing, this study included patients with evidence of multi-level disease, therefore leading to uncertainty as to which level is responsible for the observed patterns and confounding the results of the study. Currently, there are limited data on the variability with which cervical radiculopathy presents in real world situations and how often the actual presentation might deviate from the standard, textbook descriptions. Such information would be useful to surgeons making diagnoses as to a causative root level. The purposes of this study were therefore to examine cervical radiculopathy patterns in a surgical population undergoing single-level anterior cervical discectomy and fusion (ACDF) for refractory radiculopathy and to determine how often patients present with standard versus non-standard patterns. Single-level disease was chosen in order to correlate the presenting symptoms with an isolated root level.
Discussion
Figure 1: Schematic representation of the observed standard radicular patterns.
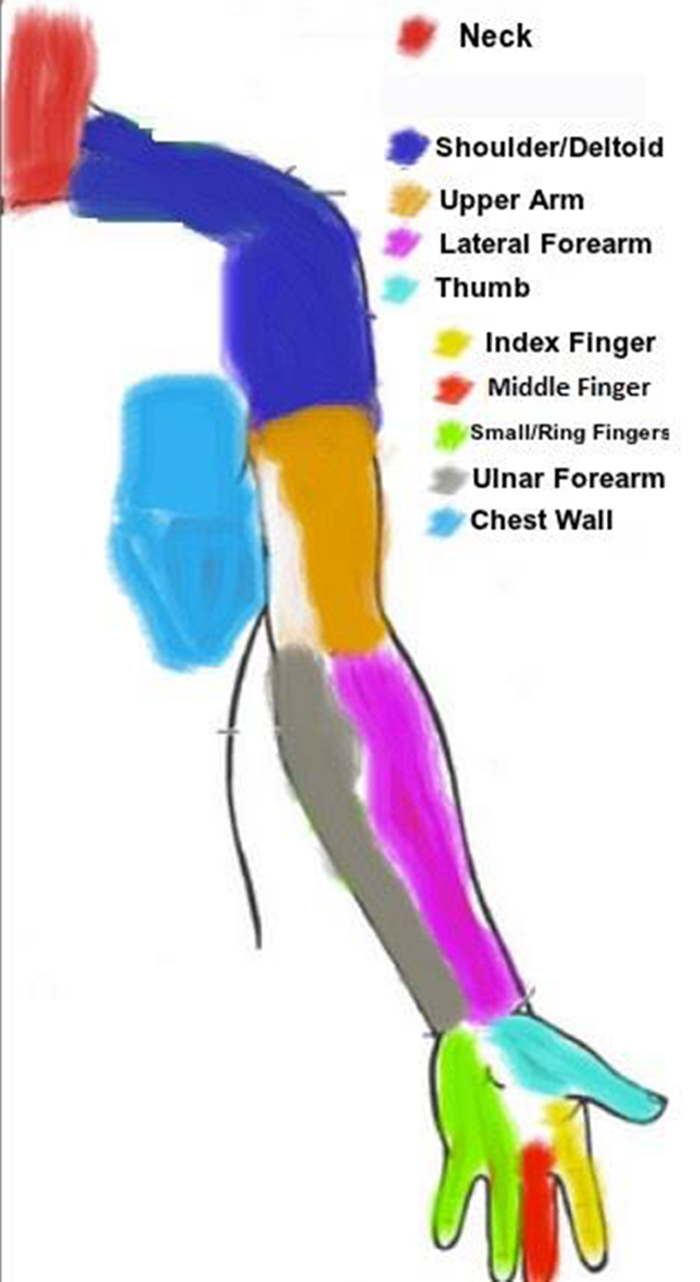
Figure 1 Traditional, textbook teaching holds that cervical radiculopathy presents with a reproducible symptom pattern according to established anatomic maps. However, the results of this study confirm that presentation in clinical practice may vary widely from these standard, expected patterns. Out of two hundred and thirty-nine patients, the observed pattern of cervical radiculopathy adhered to the standard pattern in only 54% (129/239). Furthermore, this variance was not affected by the level of the cervical pathology but occurred at every level. Proximal cervical levels often presented with distal symptoms, and distal cervical levels often presented with only proximal symptoms. It is important for practitioners to be aware not only of the standard, “Netter-diagram” patterns but also of this variability when treating patients with cervical radiculopathy, in particular when identifying operative levels in those requiring surgery.
While considerable data exists describing both nerve rootlet variability as well as brachial plexus variants, there remains a paucity of literature documenting the radicular patterns in single level cervical pathology. In the present study, when a non-standard radicular pattern was present, the variance was on average 1.68 levels from the expected pattern. Naturally occurring variants in cervical nerve rootlet anatomy is one possible explanation for this variation. Tanaka et al. [7] in a cadaveric anatomic study of the cervical nerve roots found that over 50% of the time there were intradural intersegmental connections of the dorsal rootlets between C5 and C6 and between C6 and C7. Kuba et al. [15] reported similar findings with C5–6 and C6–7 being the most common levels with rootlet anastomosis. On the basis of the variability found in their cadaveric study, Karatas et al. [11] noted that the true pathological level may be one higher or lower than what the symptoms dictate, and that careful assessment of the neurological, physiological, and radiological findings should always be undertaken.
Variability in brachial plexus anatomy can also possibly explain some of the variation in the clinical presentation of radicular symptoms. The brachial plexus is comprised of the ventral rami of the C5–T1 spinal roots. Based on anatomic dissections of the plexus, a “typical” plexus is only observed 37–77% of the time. [4–6] The two most common variants of the brachial plexus include prefixation and postfixation. Prefixation of the plexus occurs when the C4 nerve root provides considerable contribution to the plexus with the T1 nerve root providing minimal to no contribution. A postfixed plexus occurs when the plexus receives little to no contribution from the C5 nerve root and instead receives considerable innervation from the T2 nerve root. Overall, a prefixed plexus occurs more frequently than a postfixed plexus (26–48% versus 4%). [4–6, 16–19] Functionally, a prefixed or postfixed brachial plexus can shift the observed pattern of cervical radiculopathy one level cranial or caudal depending on the anatomic variant present. [20]
A recent study by Rainville et al. reported on a case series of patients with MRI confirmed C6 or C7 nerve root compression with symptoms consistent with cervical radiculopathy. [21] The authors found that arm pain symptoms in these patients were diffuse in nature and not distinctly different between those patients with C6 or C7 nerve compression. Furthermore, patterns of weakness were not significantly different between the two groups. The authors concluded that given the degree of overlap in the presentation of C6 and C7 radiculopathy, practitioners should exercise caution when predicting root involvement based on symptoms.
There was a significant difference in laterality of symptoms, with right-sided symptoms arising in 51% of the standard group versus 28% in the non-standard group. The reason for this finding cannot be determined from the present study, although a speculative etiology may include differences in the embryological development of brachial plexus and spinal cord neuroanatomy, among other factors.
Ipsilateral neck pain, interscapular pain, and pain that radiates into the shoulder occurred frequently in patients in this study, irrespective of spinal level. Ipsiplateral neck pain was found in 81% of the patients and was not found to differ by the level of spinal pathology. Similarly, shoulder pain was found in nearly 60% of the patients and also did not differ by the level of pathology. Isolated ipsilateral neck /shoulder pain without any distal radicular symptoms was less common but still observed in 19% of all patients. This presentation most commonly occurred at the C3–4 (60%) and C4–5 (33%) levels, where it could be considered consistent with the standard pattern. However, levels that are classically thought to present with symptoms distal to the shoulder, such as C5–6 and C6–7, did not do so 17% and 13% of the time in this study, presenting only with neck/trapezial/shoulder symptoms. Conversely, we found that 40% of those with C3–4 pathology had distal radicular symptoms, and 37.5% of those with C4–5 pathology had additional symptoms distal to the elbow. Neither of these findings are consistent with a standard pattern.
There are potential limitations to be considered in evaluating our results. First, this is a retrospective study that relied on electronic medical record review and patient derived pain diagrams to determine the presenting pattern of radiculopathy. Being a retrospective study, the patients were not prospectively reviewed to assess adherence to inclusion criteria, and there certainly could have been heterogeneity in interpretation of the MRI findings among the 6 treating surgeons. The MRI sequence was also not standardized in this series, as patients presented with MRIs not only from our institution but from a multitude of outside institutions, potentially affecting interpretation as well. Second, we used a 75% or greater reduction in pre-operative symptoms as an inclusion criterion for the study, with the 75% or greater improvement used as an indicator that the correct level of pathology had been addressed. However, it remains possible that radiculopathy at other levels could account for the remaining up to 25% of symptoms left unresolved in those who did not get 100% relief.
Third, it is also possible that portions of the reported symptoms may be related to pathology other than radiculopathy. For example, segmental spondylosis may contribute to neck, shoulder and trapezial symptoms. However, given the 75% post-operative improvement criterion, it remains likely that the symptoms are referable to the operative level, whether due to nerve root compression, spondylosis, or some combination of the two. Furthermore, surgery was never performed in this series for generalized, diffuse axial pain. In order to be indicated for surgery, any neck pain had to be unilateral and ipsilateral to demonstrable root compression, and it had to radiate to at least the shoulder area. Therefore, we believe the findings of this study remain relevant to clinical decision-making when ascribing symptoms to a cervical level demonstrating root compression.
In those presenting with non-standard radicular complaints, careful assessment is required to determine the causative level(s). Findings on neurologic exam. such as motor weakness, sensory abnormalities, and reflex changes, are obviously of importance, but they are limited in that not all patients with cervical radiculopathy demonstrate neurologic findings. Reproduction of symptoms with Spurling’s maneuver, if present, implies a cervical source but does not identify a causative level. Detailed physical examination of the upper extremities should be performed, including assessment for common compressive neuropathies of the elbow and wrist such as cubital or carpal tunnel syndrome. Although additional information may be gleaned from responses to selective nerve root injections and electrodiagnostic studies, there remains no clear evidence that either of these can be used reliably as predictive tests. We therefore do not routinely rely on electrodiagnotic tests when trying to identify a causative level(s) in cervical radiculopathy, although they may be helpful in evaluating non-spinal etiologies, such as peripheral neuropathy and compression (e.g., carpal tunnel, cubital tunnel, etc). In the end, identifying the causative level(s) in cervical radiculopathy remains a clinical process requiring both judgment and experience. We believe that the findings of this study will be useful when trying to reconcile non-standard symptoms with physical findings and imaging.
In conclusion, standard patterns of cervical radiculopathy were found in only 54% of patients undergoing single level ACDF. This divergence from the accepted standard highlights the need for careful assessment of associated neurological, physical, radiological, and other findings, as a non-standard clinical pattern is frequently encountered. It is not only possible, but actually relatively common, for patients with C3–4 or C4–5 radiculopathy to complain of distal symptoms that seemingly do not make “sense.” Similarly, we found that it is also relatively common for patients with C5–6 or C6–7 radiculopathy to complain only of proximal symptoms. In order to appropriately treat patients presenting with cervical radiculopathy, practitioners must think broadly when identifying causative levels because they frequently do not adhere to the accepted standard in actual clinical practice.
References:
Carette S, Fehlings MG.
Clinical practice. Cervical radiculopathy.
The New England journal of medicine 2005; 353: 392-399
DOI: 10.1056/NEJMcp043887Ellenberg MR, Honet JC, Treanor WJ.
Cervical radiculopathy.
Archives of physical medicine and rehabilitation 1994; 75: 342-352Fouyas IP, Statham PF, Sandercock PA.
Cochrane review on the role of surgery in cervical spondylotic radiculomyelopathy.
Spine 2002; 27: 736-747Lee HY, Chung IH, Sir WS et al.
Variations of the ventral rami of the brachial plexus.
J Korean Med Sci 1992; 7: 19-24
DOI: 10.3346/jkms.1992.7.1.19Matejcik V.
Variations of nerve roots of the brachial plexus.
Bratisl Lek Listy 2005; 106: 34-36Uysal, II, Seker M, Karabulut AK et al.
Brachial plexus variations in human fetuses.
Neurosurgery 2003; 53: 676-684; discussion 684Tanaka N, Fujimoto Y, An HS et al.
The anatomic relation among the nerve roots, intervertebral foramina, and intervertebral discs of the cervical spine.
Spine 2000; 25: 286-291Perneczky A, Sunder-Plassmann M.
Intradural variant of cervical nerve root fibres. Potential cause of misinterpreting the segmental location
of cervical disc prolpases from clinical evidence.
Acta Neurochir (Wien) 1980; 52: 79-83Pallie W, Manuel JK.
Intersegmental anastomoses between dorsal spinal rootlets in some vertebrates.
Acta Anat (Basel) 1968; 70: 341-351Pallie W.
The intersegmental anastomoses of posterior spinal rootlets and their significance.
J Neurosurg 1959; 16: 188-196
DOI: 10.3171/jns.1959.16.2.0188Karatas A, Caglar S, Savas A et al.
Microsurgical anatomy of the dorsal cervical rootlets and dorsal root entry zones.
Acta Neurochir (Wien) 2005; 147: 195-199; discussion 199 DOI: 10.1007/s00701-004-0425-yMurphy DR, Hurwitz EL, Gerrard JK et al.
Pain Patterns and Descriptions in Patients with Radicular Pain:
Does the Pain Necessarily Follow a Specific Dermatome?
Chiropractic & Osteopathy 2009 (Sep 21); 17 (1): 9Keegan JJ, Garrett FD.
The segmental distribution of the cutaneous nerves in the limbs of man.
Anat Rec 1948; 102: 409-437Netter FH.
Atlas of Human Anatomy. 6th ed.
Philadelphia: Saunders/Elsevier; 2015Kubo Y, Waga S, Kojima T et al.
Microsurgical anatomy of the lower cervical spine and cord.
Neurosurgery 1994; 34: 895-890; discussion 901-892Tubbs RS, Loukas M, Shoja MM et al.
Contributions of the fourth spinal nerve to the brachial plexus without prefixation.
J Neurosurg Spine 2008; 8: 548-551
DOI: 10.3171/SPI/2008/8/6/548Matejcik V.
Aberrant formation and clinical picture of brachial plexus from the point of view of a neurosurgeon.
Bratisl Lek Listy 2003; 104: 291-299Loukas M, Louis RG, Jr., Wartmann CT.
T2 contributions to the brachial plexus.
Neurosurgery 2007; 60: ONS13-18; discussion ONS18
DOI: 10.1227/01.NEU.0000249234.20484.2AJohnson EO, Vekris M, Demesticha T et al.
Neuroanatomy of the brachial plexus: normal and variant anatomy of its formation.
Surg Radiol Anat 2010; 32: 291-297
DOI: 10.1007/s00276-010-0646-0Kuzma SA, Doberstein ST, Rushlow DR.
Postfixed brachial plexus radiculopathy due to thoracic disc herniation in a collegiate wrestler: a case report.
J Athl Train 2013; 48: 710-715
DOI: 10.4085/1062-6050-48.5.03Rainville J, Joyce AA, Laxer E et al.
Comparison of Symptoms From C6 and C7 Radiculopathy.
Spine (Phila Pa 1976) 2017; 42: 1545-1551
DOI: 10.1097/BRS.0000000000002353
Return to RADICULOPATHY
Return to SPINAL PAIN MANAGEMENT
Since 6-29-2019


| Home Page | Visit Our Sponsors | Become a Sponsor |
Please read our DISCLAIMER |